Chitinase inhibition promotes atherosclerosis in hyperlipidemic mice
- PMID: 23685110
- PMCID: PMC3702742
- DOI: 10.1016/j.ajpath.2013.04.003
Chitinase inhibition promotes atherosclerosis in hyperlipidemic mice
Abstract
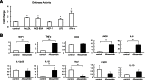
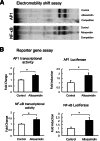
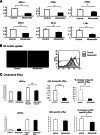
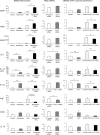
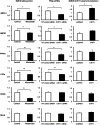
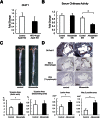
Chitinase 1 (CHIT1) is secreted by activated macrophages. Chitinase activity is raised in atherosclerotic patient sera and is present in atherosclerotic plaque. However, the role of CHIT1 in atherosclerosis is unknown. Preliminary studies of atherosclerosis in cynomolgous monkeys revealed CHIT1 to be closely correlated with areas of macrophage infiltration. Thus, we investigated the effects of a chitinase inhibitor, allosamidin, on macrophage function in vitro and on atherosclerotic development in vivo. In RAW264.7 cells, allosamidin elevated monocyte chemoattractant protein 1 and tumor necrosis factor alpha expression, and increased activator protein 1 and nuclear factor-κB transcriptional activity. Although inducible nitric oxide synthase, IL-6, and IL-1β expression were increased, Arg1 expression was decreased by chitinase inhibition, suggesting that suppression of CHIT1 activity polarizes macrophages into a M1 phenotype. Allosamidin decreased scavenger receptor AI, CD36, ABCA1, and ABCG1 expression which led to suppression of cholesterol uptake and apolipoprotein AI-mediated cholesterol efflux in macrophages. These effects were confirmed with CHIT1 siRNA transfection and CHIT1 plasmid transfection experiments in primary macrophages. Apolipoprotein E-deficient hyperlipidemic mice treated for 6 weeks with constant administration of allosamidin and fed an atherogenic diet showed aggravated atherosclerotic lesion formation. These data suggest that CHIT1 exerts protective effects against atherosclerosis by suppressing inflammatory responses and polarizing macrophages toward an M2 phenotype, and promoting lipid uptake and cholesterol efflux in macrophages.
Copyright © 2013 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures
References
-
- Libby P., Ridker P.M., Hansson G.K. Progress and challenges in translating the biology of atherosclerosis. Nature. 2011;473:317–325. - PubMed
-
- Moreno P.R., Falk E., Palacios I.F., Newell J.B., Fuster V., Fallon J.T. Macrophage infiltration in acute coronary syndromes. Implications for plaque rupture. Circulation. 1994;90:775–778. - PubMed
-
- Renkema G.H., Boot R.G., Au F.L., Donker-Koopman W.E., Strijland A., Muijsers A.O., Hrebicek M., Aerts J.M. Chitotriosidase, a chitinase, and the 39-kDa human cartilage glycoprotein, a chitin-binding lectin, are homologues of family 18 glycosyl hydrolases secreted by human macrophages. Eur J Biochem. 1998;251:504–509. - PubMed
-
- Boot R.G., van Achterberg T.A., van Aken B.E., Renkema G.H., Jacobs M.J., Aerts J.M., de Vries C.J. Strong induction of members of the chitinase family of proteins in atherosclerosis: chitotriosidase and human cartilage gp-39 expressed in lesion macrophages. Arterioscler Thromb Vasc Biol. 1999;19:687–694. - PubMed
-
- Artieda M., Cenarro A., Gañán A., Jericó I., Gonzalvo C., Casado J.M., Vitoria I., Puzo J., Pocoví M., Civeira F. Serum chitotriosidase activity is increased in subjects with atherosclerosis disease. Arterioscler Thromb Vasc Biol. 2003;23:1645–1652. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

