Proprotein convertases play an important role in regulating PKGI endoproteolytic cleavage and nuclear transport
- PMID: 23686857
- PMCID: PMC3726948
- DOI: 10.1152/ajplung.00391.2012
Proprotein convertases play an important role in regulating PKGI endoproteolytic cleavage and nuclear transport
Abstract
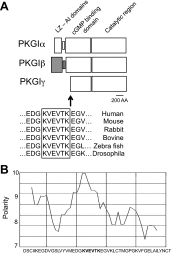
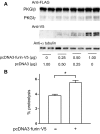
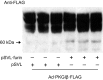
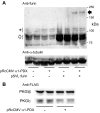
Nitric oxide and cGMP modulate vascular smooth muscle cell (SMC) phenotype by regulating cell differentiation and proliferation. Recent studies suggest that cGMP-dependent protein kinase I (PKGI) cleavage and the nuclear translocation of a constitutively active kinase fragment, PKGIγ, are required for nuclear cGMP signaling in SMC. However, the mechanisms that control PKGI proteolysis are unknown. Inspection of the amino acid sequence of a PKGI cleavage site that yields PKGIγ and a protease database revealed a putative minimum consensus sequence for proprotein convertases (PCs). Therefore we investigated the role of PCs in regulating PKGI proteolysis. We observed that overexpression of PCs, furin and PC5, but not PC7, which are all expressed in SMC, increase PKGI cleavage in a dose-dependent manner in human embryonic kidney (HEK) 293 cells. Moreover, furin-induced proteolysis of mutant PKGI, in which alanines were substituted into the putative PC consensus sequence, was decreased in these cells. In addition, overexpression of furin increased PKGI proteolysis in LoVo cells, which is an adenocarcinoma cell line expressing defective furin without PC activity. Also, expression of α1-PDX, an engineered serpin-like PC inhibitor, reduced PC activity and decreased PKGI proteolysis in HEK293 cells. Last, treatment of low-passage rat aortic SMC with membrane-permeable PC inhibitor peptides decreased cGMP-stimulated nuclear PKGIγ translocation. These data indicate for the first time that PCs have a role in regulating PKGI proteolysis and the nuclear localization of its active cleavage product, which are important for cGMP-mediated SMC phenotype.
Keywords: guanosine 3′,5′-cyclic monophosphate-dependent protein kinase I; nitric oxide and guanosine 3′,5′-cyclic monophosphate signaling; proprotein convertase.
Figures



Similar articles
-
cGMP-dependent protein kinase I gamma encodes a nuclear localization signal that regulates nuclear compartmentation and function.Cell Signal. 2014 Dec;26(12):2633-44. doi: 10.1016/j.cellsig.2014.08.004. Epub 2014 Aug 27. Cell Signal. 2014. PMID: 25172423 Free PMC article.
-
The Golgi apparatus regulates cGMP-dependent protein kinase I compartmentation and proteolysis.Am J Physiol Cell Physiol. 2015 Jun 1;308(11):C944-58. doi: 10.1152/ajpcell.00199.2014. Epub 2015 Apr 8. Am J Physiol Cell Physiol. 2015. PMID: 25855081 Free PMC article.
-
Proteolytic processing of cGMP-dependent protein kinase I mediates nuclear cGMP signaling in vascular smooth muscle cells.Circ Res. 2008 Jul 3;103(1):53-60. doi: 10.1161/CIRCRESAHA.108.176321. Epub 2008 Jun 5. Circ Res. 2008. PMID: 18535260 Free PMC article.
-
Knock-out mouse models of proprotein convertases: unique functions or redundancy?Front Biosci. 2008 May 1;13:4960-71. doi: 10.2741/3055. Front Biosci. 2008. PMID: 18508561 Review.
-
The proprotein convertases furin and PACE4 play a significant role in tumor progression.Mol Carcinog. 2000 Jun;28(2):63-9. Mol Carcinog. 2000. PMID: 10900462 Review.
Cited by
-
Furin Regulates the Alveolarization of Neonatal Lungs in a Mouse Model of Hyperoxic Lung Injury.Biomolecules. 2023 Nov 16;13(11):1656. doi: 10.3390/biom13111656. Biomolecules. 2023. PMID: 38002338 Free PMC article.
-
Transforming growth factor-β downregulates sGC subunit expression in pulmonary artery smooth muscle cells via MEK and ERK signaling.Am J Physiol Lung Cell Mol Physiol. 2019 Jan 1;316(1):L20-L34. doi: 10.1152/ajplung.00319.2018. Epub 2018 Sep 27. Am J Physiol Lung Cell Mol Physiol. 2019. PMID: 30260287 Free PMC article.
-
N-Linked Glycosylation-Dependent and -Independent Mechanisms Regulating CTRP12 Cleavage, Secretion, and Stability.Biochemistry. 2019 Feb 12;58(6):727-741. doi: 10.1021/acs.biochem.8b00528. Epub 2019 Jan 4. Biochemistry. 2019. PMID: 30566828 Free PMC article.
-
cGMP-dependent protein kinase I gamma encodes a nuclear localization signal that regulates nuclear compartmentation and function.Cell Signal. 2014 Dec;26(12):2633-44. doi: 10.1016/j.cellsig.2014.08.004. Epub 2014 Aug 27. Cell Signal. 2014. PMID: 25172423 Free PMC article.
-
The Golgi apparatus regulates cGMP-dependent protein kinase I compartmentation and proteolysis.Am J Physiol Cell Physiol. 2015 Jun 1;308(11):C944-58. doi: 10.1152/ajpcell.00199.2014. Epub 2015 Apr 8. Am J Physiol Cell Physiol. 2015. PMID: 25855081 Free PMC article.
References
-
- Anderson ED, Thomas L, Hayflick JS, Thomas G. Inhibition of HIV-1 gp160-dependent membrane fusion by a furin-directed alpha 1-antitrypsin variant. J Biol Chem 268: 24887–24891, 1993 - PubMed
-
- Artenstein AW, Opal SM. Proprotein convertases in health and disease. N Engl J Med 365: 2507–2518, 2011 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

