Efficacy, safety, pharmacokinetics and biomarker findings in patients with HER2-positive advanced or metastatic breast cancer treated with lapatinib in combination with capecitabine: results from 51 Japanese patients treated in a clinical study
- PMID: 23689990
- PMCID: PMC4331616
- DOI: 10.1007/s12282-013-0475-1
Efficacy, safety, pharmacokinetics and biomarker findings in patients with HER2-positive advanced or metastatic breast cancer treated with lapatinib in combination with capecitabine: results from 51 Japanese patients treated in a clinical study
Abstract
Background: The results from a phase III trial conducted outside of Japan demonstrated a significant improvement in time to progression (TTP) when lapatinib was combined with capecitabine compared with capecitabine alone in patients with HER2-positive advanced or metastatic breast cancer. In this clinical study of lapatinib in combination with capecitabine, efficacy, safety, pharmacokinetics (PK) and biomarkers were investigated in Japanese patients with HER2-positive advanced or metastatic breast cancer treated with prior trastuzumab.
Methods: Eligible women received lapatinib 1250 mg once daily and capecitabine 1000 mg/m(2) twice daily on days 1 through 14 of a 21-day cycle. The primary endpoint was the clinical benefit rate (CBR: complete response, partial response or stable disease for at least 24 weeks).
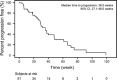
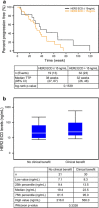
Results: Lapatinib in combination with capecitabine was well tolerated in the 51 patients enrolled in this study. CBR was 59 % (95 % CI 44.2, 72.4), and the median TTP in the Kaplan-Meier estimate was 36 weeks (95 % CI 27.1, 48.0). The majority of drug-related adverse events were mild to moderate (grade 1 or 2); the most common adverse events reported were palmar-plantar erythrodysesthesia syndrome (76 %), diarrhea (67 %) and stomatitis (41 %).
Conclusions: Lapatinib in combination with capecitabine in Japanese HER2-positive breast cancer patients was well tolerated. Overall, our findings on the efficacy, safety and PK were similar to those reported from the overseas studies.
Figures
Similar articles
-
Trastuzumab emtansine versus capecitabine plus lapatinib in patients with previously treated HER2-positive advanced breast cancer (EMILIA): a descriptive analysis of final overall survival results from a randomised, open-label, phase 3 trial.Lancet Oncol. 2017 Jun;18(6):732-742. doi: 10.1016/S1470-2045(17)30312-1. Epub 2017 May 16. Lancet Oncol. 2017. PMID: 28526536 Free PMC article. Clinical Trial.
-
Safety and efficacy of neratinib in combination with capecitabine in patients with metastatic human epidermal growth factor receptor 2-positive breast cancer.J Clin Oncol. 2014 Nov 10;32(32):3626-33. doi: 10.1200/JCO.2014.56.3809. Epub 2014 Oct 6. J Clin Oncol. 2014. PMID: 25287822 Clinical Trial.
-
Lapatinib plus capecitabine in treating HER2-positive advanced breast cancer: efficacy, safety, and biomarker results from Chinese patients.Chin J Cancer. 2011 May;30(5):327-35. doi: 10.5732/cjc.010.10507. Chin J Cancer. 2011. PMID: 21527065 Free PMC article. Clinical Trial.
-
Lapatinib: a dual inhibitor of human epidermal growth factor receptor tyrosine kinases.Clin Ther. 2008 Aug;30(8):1426-47. doi: 10.1016/j.clinthera.2008.08.008. Clin Ther. 2008. PMID: 18803986 Review.
-
Lapatinib: a small-molecule inhibitor of epidermal growth factor receptor and human epidermal growth factor receptor-2 tyrosine kinases used in the treatment of breast cancer.Clin Ther. 2009;31 Pt 2:2332-48. doi: 10.1016/j.clinthera.2009.11.029. Clin Ther. 2009. PMID: 20110044 Review.
Cited by
-
Cost-effectiveness analysis of adjuvant chemotherapies in patients presenting with gastric cancer after D2 gastrectomy.BMC Cancer. 2014 Dec 19;14:984. doi: 10.1186/1471-2407-14-984. BMC Cancer. 2014. PMID: 25526802 Free PMC article.
-
BioPATH: A Biomarker Study in Asian Patients with HER2+ Advanced Breast Cancer Treated with Lapatinib and Other Anti-HER2 Therapy.Cancer Res Treat. 2019 Oct;51(4):1527-1539. doi: 10.4143/crt.2018.598. Epub 2019 Jun 4. Cancer Res Treat. 2019. PMID: 31163957 Free PMC article.
-
Chemotherapy protocols and incidence of oral mucositis. An integrative review.Einstein (Sao Paulo). 2018;16(1):eRW4007. doi: 10.1590/s1679-45082018rw4007. Epub 2018 Apr 23. Einstein (Sao Paulo). 2018. PMID: 29694618 Free PMC article. Review.
-
Efficacy and safety of lapatinib and trastuzumab for HER2-positive breast cancer: a systematic review and meta-analysis of randomised controlled trials.BMJ Open. 2017 Mar 13;7(3):e013053. doi: 10.1136/bmjopen-2016-013053. BMJ Open. 2017. PMID: 28289045 Free PMC article.
-
Efficacy and mechanism of action of the tyrosine kinase inhibitors gefitinib, lapatinib and neratinib in the treatment of HER2-positive breast cancer: preclinical and clinical evidence.Am J Cancer Res. 2015 Aug 15;5(9):2531-61. eCollection 2015. Am J Cancer Res. 2015. PMID: 26609467 Free PMC article. Review.
References
-
- Herbst RS, Maddox AM, Rothenberg ML, Small EJ, Rubin EH, Baselga J, et al. Selective oral epidermal growth factor receptor tyrosine kinase inhibitor ZD1839 is generally well-tolerated and has activity in non-small-cell lung cancer and other solid tumors: results of a phase I trial. J Clin Oncol. 2002;20(18):3815–3825. doi: 10.1200/JCO.2002.03.038. - DOI - PubMed
-
- Cameron D, Casey M, Press M, Lindquist D, Pienkowski T, Romieu CG, et al. A phase III randomized comparison of lapatinib plus capecitabine versus capecitabine alone in women with advanced breast cancer that has progressed on trastuzumab: updated efficacy and biomarker analyses. Breast Cancer Res Treat. 2008;112(3):533–543. doi: 10.1007/s10549-007-9885-0. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

