Carotid body hyperplasia and enhanced ventilatory responses to hypoxia in mice with heterozygous deficiency of PHD2
- PMID: 23690557
- PMCID: PMC3731614
- DOI: 10.1113/jphysiol.2012.247254
Carotid body hyperplasia and enhanced ventilatory responses to hypoxia in mice with heterozygous deficiency of PHD2
Abstract
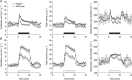
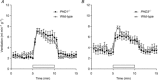
Oxygen-dependent prolyl hydroxylation of hypoxia-inducible factor (HIF) by a set of closely related prolyl hydroxylase domain enzymes (PHD1, 2 and 3) regulates a range of transcriptional responses to hypoxia. This raises important questions about the role of these oxygen-sensing enzymes in integrative physiology. We investigated the effect of both genetic deficiency and pharmacological inhibition on the change in ventilation in response to acute hypoxic stimulation in mice. Mice exposed to chronic hypoxia for 7 days manifest an exaggerated hypoxic ventilatory response (HVR) (10.8 ± 0.3 versus 4.1 ± 0.7 ml min(-1) g(-1) in controls; P < 0.01). HVR was similarly exaggerated in PHD2(+/-) animals compared to littermate controls (8.4 ± 0.7 versus 5.0 ± 0.8 ml min(-1) g(-1); P < 0.01). Carotid body volume increased (0.0025 ± 0.00017 in PHD2(+/-) animals versus 0.0015 ± 0.00019 mm(3) in controls; P < 0.01). In contrast, HVR in PHD1(-/-) and PHD3(-/-) mice was similar to littermate controls. Acute exposure to a small molecule PHD inhibitor (PHI) (2-(1-chloro-4-hydroxyisoquinoline-3-carboxamido) acetic acid) did not mimic the ventilatory response to hypoxia. Further, 7 day administration of the PHI induced only modest increases in HVR and carotid body cell proliferation, despite marked stimulation of erythropoiesis. This was in contrast with chronic hypoxia, which elicited both exaggerated HVR and cellular proliferation. The findings demonstrate that PHD enzymes modulate ventilatory sensitivity to hypoxia and identify PHD2 as the most important enzyme in this response. They also reveal differences between genetic inactivation of PHDs, responses to hypoxia and responses to a pharmacological inhibitor, demonstrating the need for caution in predicting the effects of therapeutic modulation of the HIF hydroxylase system on different physiological responses.
Figures





References
-
- Appelhoff RJ, Tian YM, Raval RR, Turley H, Harris AL, Pugh CW, Ratcliffe PJ, Gleadle JM. Differential function of the prolyl hydroxylases PHD1, PHD2, and PHD3 in the regulation of hypoxia-inducible factor. J Biol Chem. 2004;279:38458–38465. - PubMed
-
- Aragones J, Schneider M, Van Geyte K, Fraisl P, Dresselaers T, Mazzone M, Dirkx R, Zacchigna S, Lemieux H, Jeoung NH, Lambrechts D, Bishop T, Lafuste P, Diez-Juan A, Harten SK, Van Noten P, De Bock K, Willam C, Tjwa M, Grosfeld A, Navet R, Moons L, Vandendriessche T, Deroose C, Wijeyekoon B, Nuyts J, Jordan B, Silasi-Mansat R, Lupu F, Dewerchin M, Pugh C, Salmon P, Mortelmans L, Gallez B, Gorus F, Buyse J, Sluse F, Harris RA, Gnaiger E, Hespel P, Van Hecke P, Schuit F, Van Veldhoven P, Ratcliffe P, Baes M, Maxwell P, Carmeliet P. Deficiency or inhibition of oxygen sensor Phd1 induces hypoxia tolerance by reprogramming basal metabolism. Nat Genet. 2008;40:170–180. - PubMed
-
- Bishop T, Gallagher D, Pascual A, Lygate CA, de Bono JP, Nicholls LG, Ortega-Saenz P, Oster H, Wijeyekoon B, Sutherland AI, Grosfeld A, Aragones J, Schneider M, van Geyte K, Teixeira D, Diez-Juan A, Lopez-Barneo J, Channon KM, Maxwell PH, Pugh CW, Davies AM, Carmeliet P, Ratcliffe PJ. Abnormal sympathoadrenal development and systemic hypotension in PHD3−/− mice. Mol Cell Biol. 2008;28:3386–3400. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

