PI3K-Akt signaling activates mTOR-mediated epileptogenesis in organotypic hippocampal culture model of post-traumatic epilepsy
- PMID: 23699517
- PMCID: PMC3735438
- DOI: 10.1523/JNEUROSCI.3870-12.2013
PI3K-Akt signaling activates mTOR-mediated epileptogenesis in organotypic hippocampal culture model of post-traumatic epilepsy
Abstract
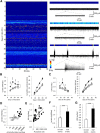
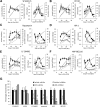
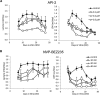
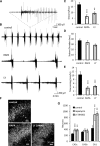
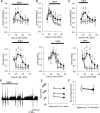
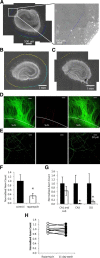
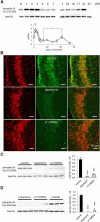
mTOR is activated in epilepsy, but the mechanisms of mTOR activation in post-traumatic epileptogenesis are unknown. It is also not clear whether mTOR inhibition has an anti-epileptogenic, or merely anticonvulsive effect. The rat hippocampal organotypic culture model of post-traumatic epilepsy was used to study the effects of long-term (four weeks) inhibition of signaling pathways that interact with mTOR. Ictal activity was quantified by measurement of lactate production and electrical recordings, and cell death was quantified with lactate dehydrogenase (LDH) release measurements and Nissl-stained neuron counts. Lactate and LDH measurements were well correlated with electrographic activity and neuron counts, respectively. Inhibition of PI3K and Akt prevented activation of mTOR, and was as effective as inhibition of mTOR in reducing ictal activity and cell death. A dual inhibitor of PI3K and mTOR, NVP-BEZ235, was also effective. Inhibition of mTOR with rapamycin reduced axon sprouting. Late start of rapamycin treatment was effective in reducing epileptic activity and cell death, while early termination of rapamycin treatment did not result in increased epileptic activity or cell death. The conclusions of the study are as follows: (1) the organotypic hippocampal culture model of post-traumatic epilepsy comprises a rapid assay of anti-epileptogenic and neuroprotective activities and, in this model (2) mTOR activation depends on PI3K-Akt signaling, and (3) transient inhibition of mTOR has sustained effects on epilepsy.
Figures
Similar articles
-
Interleukin-1β plays a role in the pathogenesis of mesial temporal lobe epilepsy through the PI3K/Akt/mTOR signaling pathway in hippocampal neurons.J Neuroimmunol. 2015 May 15;282:110-7. doi: 10.1016/j.jneuroim.2015.04.003. Epub 2015 Apr 2. J Neuroimmunol. 2015. PMID: 25903737
-
The effect of IL-1β on synaptophysin expression and electrophysiology of hippocampal neurons through the PI3K/Akt/mTOR signaling pathway in a rat model of mesial temporal lobe epilepsy.Neurol Res. 2017 Jul;39(7):640-648. doi: 10.1080/01616412.2017.1312070. Epub 2017 Apr 4. Neurol Res. 2017. PMID: 28372486
-
miR-10a induces inflammatory responses in epileptic hippocampal neurons of rats via PI3K/Akt/mTOR signaling pathway.Neuroreport. 2023 Jun 7;34(10):526-534. doi: 10.1097/WNR.0000000000001920. Epub 2023 May 20. Neuroreport. 2023. PMID: 37270844
-
Therapeutic role of targeting mTOR signaling and neuroinflammation in epilepsy.Epilepsy Res. 2020 Mar;161:106282. doi: 10.1016/j.eplepsyres.2020.106282. Epub 2020 Jan 30. Epilepsy Res. 2020. PMID: 32036255 Free PMC article. Review.
-
An insight into crosstalk among multiple signaling pathways contributing to epileptogenesis.Eur J Pharmacol. 2021 Nov 5;910:174469. doi: 10.1016/j.ejphar.2021.174469. Epub 2021 Sep 1. Eur J Pharmacol. 2021. PMID: 34478688 Review.
Cited by
-
mTOR inhibition in epilepsy: rationale and clinical perspectives.CNS Drugs. 2015 Feb;29(2):91-9. doi: 10.1007/s40263-014-0223-x. CNS Drugs. 2015. PMID: 25633849 Free PMC article. Review.
-
Socrates: A Novel N-Ethyl-N-nitrosourea-Induced Mouse Mutant with Audiogenic Epilepsy.Int J Mol Sci. 2023 Dec 4;24(23):17104. doi: 10.3390/ijms242317104. Int J Mol Sci. 2023. PMID: 38069426 Free PMC article.
-
Association of CDKN2A alterations with increased postoperative seizure risk after resection of brain metastases.Neurosurg Focus. 2023 Aug;55(2):E14. doi: 10.3171/2023.5.FOCUS23133. Neurosurg Focus. 2023. PMID: 37527678 Free PMC article.
-
Epileptogenesis.Cold Spring Harb Perspect Med. 2015 Sep 18;5(10):a022822. doi: 10.1101/cshperspect.a022822. Cold Spring Harb Perspect Med. 2015. PMID: 26385090 Free PMC article. Review.
-
Post-Traumatic Epilepsy and Comorbidities: Advanced Models, Molecular Mechanisms, Biomarkers, and Novel Therapeutic Interventions.Pharmacol Rev. 2022 Apr;74(2):387-438. doi: 10.1124/pharmrev.121.000375. Pharmacol Rev. 2022. PMID: 35302046 Free PMC article. Review.
References
-
- Bonfoco E, Krainc D, Ankarcrona M, Nicotera P, Lipton SA. Apoptosis and necrosis: two distinct events induced, respectively, by mild and intense insults with N-methyl-D-aspartate or nitric oxide/superoxide in cortical cell cultures. Proc Natl Acad Sci U S A. 1995;92:7162–7166. doi: 10.1073/pnas.92.16.7162. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
