Necl-4/SynCAM-4 is expressed in myelinating oligodendrocytes but not required for axonal myelination
- PMID: 23700466
- PMCID: PMC3659047
- DOI: 10.1371/journal.pone.0064264
Necl-4/SynCAM-4 is expressed in myelinating oligodendrocytes but not required for axonal myelination
Abstract
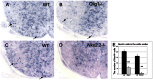
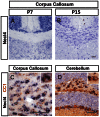
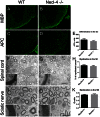
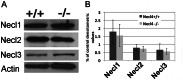
The timing and progression of axonal myelination are precisely controlled by intercellular interactions between neurons and glia in development. Previous in vitro studies demonstrated that Nectin like 4 (Necl-4, also known as cell adhesion molecule Cadm-4 or SynCAM-4) plays an essential role in axonal myelination by Schwann cells in the peripheral nervous system (PNS). However, the role of Necl-4 protein in axonal myelination in the developing central nervous system (CNS) has remained unknown. In this study, we discovered upregulation of Necl-4 expression in mature oligodendrocytes at perinatal stages when axons undergo active myelination. We generated Necl4 gene knockout mice, but found that disruption of Necl-4 gene did not affect oligodendrocyte differentiation and myelin formation in the CNS. Surprisingly, disruption of Necl-4 had no significant effect on axonal myelination in the PNS either. Therefore, our results demonstrated that Necl-4 is dispensable for axonal myelination in the developing nervous system.
Conflict of interest statement
Figures



Similar articles
-
Disruption of Nectin-like 1 cell adhesion molecule leads to delayed axonal myelination in the CNS.J Neurosci. 2008 Nov 26;28(48):12815-9. doi: 10.1523/JNEUROSCI.2665-08.2008. J Neurosci. 2008. PMID: 19036974 Free PMC article.
-
Differential Contribution of Cadm1-Cadm3 Cell Adhesion Molecules to Peripheral Myelinated Axons.J Neurosci. 2021 Feb 17;41(7):1393-1400. doi: 10.1523/JNEUROSCI.2736-20.2020. Epub 2021 Jan 4. J Neurosci. 2021. PMID: 33397712 Free PMC article.
-
A central role for Necl4 (SynCAM4) in Schwann cell-axon interaction and myelination.Nat Neurosci. 2007 Jul;10(7):861-9. doi: 10.1038/nn1915. Epub 2007 Jun 3. Nat Neurosci. 2007. PMID: 17558405 Free PMC article.
-
Promoting axonal myelination for improving neurological recovery in spinal cord injury.J Neurotrauma. 2009 Oct;26(10):1847-56. doi: 10.1089/neu.2008.0551. J Neurotrauma. 2009. PMID: 19785544 Review.
-
Nerve impulses regulate myelination through purinergic signalling.Novartis Found Symp. 2006;276:148-58; discussion 158-61, 233-7, 275-81. Novartis Found Symp. 2006. PMID: 16805428 Review.
Cited by
-
Diazoxide promotes oligodendrocyte differentiation in neonatal brain in normoxia and chronic sublethal hypoxia.Brain Res. 2014 Oct 24;1586:64-72. doi: 10.1016/j.brainres.2014.08.046. Epub 2014 Aug 23. Brain Res. 2014. PMID: 25157906 Free PMC article.
-
Tmeff2 is expressed in differentiating oligodendrocytes but dispensable for their differentiation in vivo.Sci Rep. 2017 Mar 23;7(1):337. doi: 10.1038/s41598-017-00407-1. Sci Rep. 2017. PMID: 28336932 Free PMC article.
-
Coordinated internodal and paranodal adhesion controls accurate myelination by oligodendrocytes.J Cell Biol. 2019 Sep 2;218(9):2887-2895. doi: 10.1083/jcb.201906099. Epub 2019 Aug 26. J Cell Biol. 2019. PMID: 31451613 Free PMC article.
-
Aging-related changes of optic nerve of Wistar albino rats.Age (Dordr). 2014 Apr;36(2):519-32. doi: 10.1007/s11357-013-9580-5. Epub 2013 Sep 1. Age (Dordr). 2014. PMID: 23996059 Free PMC article.
-
Neonatal hypoxia leads to impaired intestinal function and changes in the composition and metabolism of its microbiota.Sci Rep. 2025 May 1;15(1):15285. doi: 10.1038/s41598-025-00041-2. Sci Rep. 2025. PMID: 40312410 Free PMC article.
References
-
- Miller RH (2002) Regulation of oligodendrocyte development in the vertebrate CNS. Progress in Neurobiology 67: 451–467. - PubMed
-
- Rowitch DH (2004) GLIAL SPECIFICATION IN THE VERTEBRATE NEURAL TUBE. Nature Reviews Neuroscience 5: 409–419. - PubMed
-
- Baumann N, Pham-Dinh D (2001) Biology of Oligodendrocyte and Myelin in the Mammalian Central Nervous System. Physiol Rev 81: 871–927. - PubMed
-
- Simons M, Trajkovic K (2006) Neuron-glia communication in the control of oligodendrocyte function and myelin biogenesis. J Cell Sci 119: 4381–4389. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

