Chondrogenic capacity and alterations in hyaluronan synthesis of cultured human osteoarthritic chondrocytes
- PMID: 23702485
- PMCID: PMC3728838
- DOI: 10.1016/j.bbrc.2013.05.052
Chondrogenic capacity and alterations in hyaluronan synthesis of cultured human osteoarthritic chondrocytes
Abstract
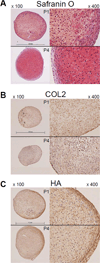
During osteoarthritis there is a disruption and loss of the extracellular matrix of joint cartilage, composed primarily of type II collagen, aggrecan and hyaluronan. In young patients, autologous chondrocyte implantation can be used to repair cartilage defects. However, for more elderly patients with osteoarthritis, such a repair approach is contraindicated because the procedure requires a large expansion of autologous chondrocytes in vitro leading a rapid, perhaps irreversible, loss of the chondrocyte phenotype. This study investigates whether osteoarthritic chondrocytes obtained from older patients can be expanded in vitro and moreover, induced to re-activate their chondrocyte phenotype. A decrease in chondrocyte phenotype markers, collagen II, aggrecan and SOX9 mRNA was observed with successive expansion of cells in monolayer culture. However, chondrogenic induction in three-dimensional pellet culture successfully rescued the expression of all three marker genes to native levels, even with 4th passage cells-cells representing an approximate 625-fold expansion in cell number. This data supports the use of osteoarthritic cells for autologous implantation repair. In addition, another set of gene products were explored as useful markers of the chondrocyte phenotype. Differentiated primary chondrocytes exhibited a common pattern of hyaluronan synthase isoforms that changed upon cell expansion in vitro and, reverted back to the original pattern following pellet culture. Moreover, the change in isoform pattern correlated with changes in the molecular size of synthesized hyaluronan.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures


References
-
- Knudson CB, Knudson W. Hyaluronan and CD44: modulators of chondrocyte metabolism. Clin Orthop Relat Res. 2004;427:S152–S162. - PubMed
-
- Nishida Y, Knudson CB, Nietfeld JJ, et al. Antisense inhibition of hyaluronan synthase-2 in human articular chondrocytes inhibits proteoglycan retention and matrix assembly. J Biol Chem. 1999;274:21893–21899. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

