Tissue plasminogen activator contributes to morphine tolerance and induces mechanical allodynia via astrocytic IL-1β and ERK signaling in the spinal cord of mice
- PMID: 23707980
- PMCID: PMC3722295
- DOI: 10.1016/j.neuroscience.2013.05.018
Tissue plasminogen activator contributes to morphine tolerance and induces mechanical allodynia via astrocytic IL-1β and ERK signaling in the spinal cord of mice
Abstract
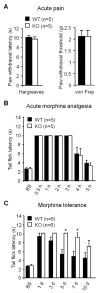
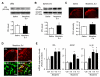
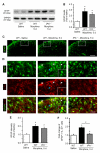
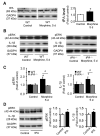
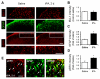
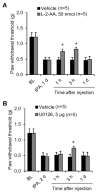
Accumulating evidence indicates that activation of spinal cord astrocytes contributes importantly to nerve injury and inflammation-induced persistent pain and chronic opioid-induced antinociceptive tolerance. Phosphorylation of extracellular signal-regulated kinase (pERK) and induction of interleukin-1 beta (IL-1β) in spinal astrocytes have been implicated in astrocytes-mediated pain. Tissue plasminogen activator (tPA) is a serine protease that has been extensively used to treat stroke. We examined the potential involvement of tPA in chronic opioid-induced antinociceptive tolerance and activation of spinal astrocytes using tPA knockout (tPA(-/-)) mice and astrocyte cultures. tPA(-/-) mice exhibited unaltered nociceptive pain and morphine-induced acute analgesia. However, the antinociceptive tolerance, induced by chronic morphine (10mg/kg/day, s.c.), is abrogated in tPA(-/-) mice. Chronic morphine induces tPA expression in glial fibrillary acidic protein (GFAP)-expressing spinal cord astrocytes. Chronic morphine also increases IL-1β expression in GFAP-expressing astrocytes, which is abolished in tPA-deficient mice. In cultured astrocytes, morphine treatment increases tPA, IL-1β, and pERK expression, and the increased IL-1β and pERK expression is abolished in tPA-deficient astrocytes. tPA is also sufficient to induce IL-1β and pERK expression in astrocyte cultures. Intrathecal injection of tPA results in up-regulation of GFAP and pERK in spinal astrocytes but not up-regulation of ionized calcium binding adapter molecule 1 in spinal microglia. Finally, intrathecal tPA elicits persistent mechanical allodynia, which is inhibited by the astroglial toxin alpha-amino adipate and the MEK (ERK kinase) inhibitor U0126. Collectively, these data suggest an important role of tPA in regulating astrocytic signaling, pain hypersensitivity, and morphine tolerance.
Keywords: DRGs; ERK; ERK kinase; FBS; GAPDH; GFAP; IBA-1; IL-1β; L-2-AA; LTP; MEK; MMP-2; PWL; WT; acute opioid analgesia; chronic morphine exposure; dorsal root ganglia; extracellular signal-regulated kinase; extracellular signal-regulated kinase (ERK); fetal bovine serum; glial fibrillary acidic protein; glyceraldehyde3-phosphate dehydrogenase; interleukin-1 beta; interleukin-1 beta (IL-1β); ionized calcium binding adapter molecule 1; l-2-Aminoadipic acid; long-term potentiation; matrix metalloprotease-2; pERK; paw withdrawal latency; phosphoERK; protease; tPA; tPA knockout mice; tissue plasminogen activator; wild-type.
Copyright © 2013 IBRO. Published by Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Acute morphine activates satellite glial cells and up-regulates IL-1β in dorsal root ganglia in mice via matrix metalloprotease-9.Mol Pain. 2012 Mar 22;8:18. doi: 10.1186/1744-8069-8-18. Mol Pain. 2012. PMID: 22439811 Free PMC article.
-
Contribution of spinal cord glial cells to L. amazonensis experimental infection-induced pain in BALB/c mice.J Neuroinflammation. 2019 May 28;16(1):113. doi: 10.1186/s12974-019-1496-2. J Neuroinflammation. 2019. PMID: 31138231 Free PMC article.
-
Spinal astrocytic activation contributes to both induction and maintenance of pituitary adenylate cyclase-activating polypeptide type 1 receptor-induced long-lasting mechanical allodynia in mice.Mol Pain. 2016 May 12;12:1744806916646383. doi: 10.1177/1744806916646383. Print 2016. Mol Pain. 2016. PMID: 27175011 Free PMC article.
-
Mechanism of allodynia evoked by intrathecal morphine-3-glucuronide in mice.Int Rev Neurobiol. 2009;85:207-19. doi: 10.1016/S0074-7742(09)85016-2. Int Rev Neurobiol. 2009. PMID: 19607972 Review.
-
Microglia: a promising target for treating neuropathic and postoperative pain, and morphine tolerance.J Formos Med Assoc. 2011 Aug;110(8):487-94. doi: 10.1016/S0929-6646(11)60074-0. J Formos Med Assoc. 2011. PMID: 21783017 Free PMC article. Review.
Cited by
-
Addressing opioid tolerance and opioid-induced hypersensitivity: Recent developments and future therapeutic strategies.Pharmacol Res Perspect. 2021 May;9(3):e00789. doi: 10.1002/prp2.789. Pharmacol Res Perspect. 2021. PMID: 34096178 Free PMC article. Review.
-
Curcumin ameliorates neuropathic pain by down-regulating spinal IL-1β via suppressing astroglial NALP1 inflammasome and JAK2-STAT3 signalling.Sci Rep. 2016 Jul 6;6:28956. doi: 10.1038/srep28956. Sci Rep. 2016. PMID: 27381056 Free PMC article.
-
Magnetic field distribution modulation of intrathecal delivered ketorolac iron-oxide nanoparticle conjugates produce excellent analgesia for chronic inflammatory pain.J Nanobiotechnology. 2018 May 16;16(1):49. doi: 10.1186/s12951-018-0375-9. J Nanobiotechnology. 2018. PMID: 29769077 Free PMC article.
-
Opioid Modulation of the Gut-Brain Axis in Opioid-Associated Comorbidities.Cold Spring Harb Perspect Med. 2021 Sep 1;11(9):a040485. doi: 10.1101/cshperspect.a040485. Cold Spring Harb Perspect Med. 2021. PMID: 32816876 Free PMC article. Review.
-
The analgesic effect and possible mechanisms by which koumine alters type II collagen-induced arthritis in rats.J Nat Med. 2019 Jan;73(1):217-225. doi: 10.1007/s11418-018-1229-3. Epub 2018 Jul 27. J Nat Med. 2019. PMID: 30054785
References
-
- Bahi A, Dreyer JL. Overexpression of plasminogen activators in the nucleus accumbens enhances cocaine-, amphetamine- and morphine-induced reward and behavioral sensitization. Genes Brain Behav. 2008;7:244–256. - PubMed
-
- Baranes D, Lederfein D, Huang YY, Chen M, Bailey CH, Kandel ER. Tissue plasminogen activator contributes to the late phase of LTP and to synaptic growth in the hippocampal mossy fiber pathway. Neuron. 1998;21:813–825. - PubMed
-
- Bessler H, Shavit Y, Mayburd E, Smirnov G, Beilin B. Postoperative pain, morphine consumption, and genetic polymorphism of IL-1beta and IL-1 receptor antagonist. Neurosci Lett. 2006;404:154–158. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

