Identification of dynamic changes in proteins associated with the cellular cytoskeleton after exposure to okadaic acid
- PMID: 23708184
- PMCID: PMC3721204
- DOI: 10.3390/md11061763
Identification of dynamic changes in proteins associated with the cellular cytoskeleton after exposure to okadaic acid
Abstract
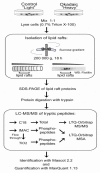
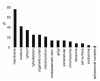
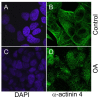
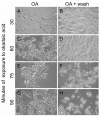
Exposure of cells to the diarrhetic shellfish poison, okadaic acid, leads to a dramatic reorganization of cytoskeletal architecture and loss of cell-cell contact. When cells are exposed to high concentrations of okadaic acid (100-500 nM), the morphological rearrangement is followed by apoptotic cell death. Okadaic acid inhibits the broad acting Ser/Thr protein phosphatases 1 and 2A, which results in hyperphosphorylation of a large number of proteins. Some of these hyperphosphorylated proteins are most likely key players in the reorganization of the cell morphology induced by okadaic acid. We wanted to identify these phosphoproteins and searched for them in the cellular lipid rafts, which have been found to contain proteins that regulate cytoskeletal dynamics and cell adhesion. By using stable isotope labeling by amino acids in cell culture cells treated with okadaic acid (400 nM) could be combined with control cells before the isolation of lipid rafts. Protein phosphorylation events and translocations induced by okadaic acid were identified by mass spectrometry. Okadaic acid was shown to regulate the phosphorylation status and location of proteins associated with the actin cytoskeleton, microtubules and cell adhesion structures. A large number of these okadaic acid-regulated proteins have previously also been shown to be similarly regulated prior to cell proliferation and migration. Our results suggest that okadaic acid activates general cell signaling pathways that induce breakdown of the cortical actin cytoskeleton and cell detachment.
Figures



References
-
- Suganuma M., Fujiki H., Suguri H., Yoshizawa S., Hirota M., Nakayasu M., Ojika M., Wakamatsu K., Yamada K., Sugimura T. Okadaic acid: An additional non-phorbol-12-tetradecanoate-13-acetate-type tumor promoter. Proc. Natl. Acad. Sci. USA. 1988;85:1768–1771. doi: 10.1073/pnas.85.6.1768. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

