Major Histocompatibility Class II Pathway Is Not Required for the Development of Nonalcoholic Fatty Liver Disease in Mice
- PMID: 23710178
- PMCID: PMC3655579
- DOI: 10.1155/2013/972962
Major Histocompatibility Class II Pathway Is Not Required for the Development of Nonalcoholic Fatty Liver Disease in Mice
Abstract
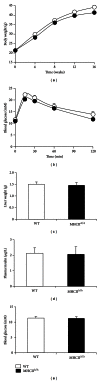
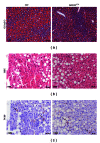
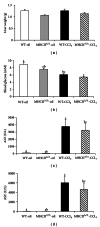
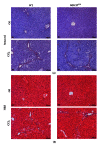
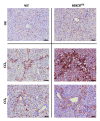
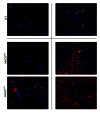
Single-nucleotide polymorphisms within major histocompatibility class II (MHC II) genes have been associated with an increased risk of drug-induced liver injury. However, it has never been addressed whether the MHC II pathway plays an important role in the development of nonalcoholic fatty liver disease, the most common form of liver disease. We used a mouse model that has a complete knockdown of genes in the MHC II pathway (MHCII(Δ/Δ)). Firstly we studied the effect of high-fat diet-induced hepatic inflammation in these mice. Secondly we studied the development of carbon-tetra-chloride- (CCl4-) induced hepatic cirrhosis. After the high-fat diet, both groups developed obesity and hepatic steatosis with a similar degree of hepatic inflammation, suggesting no impact of the knockdown of MHC II on high-fat diet-induced inflammation in mice. In the second study, we confirmed that the CCl4 injection significantly upregulated the MHC II genes in wild-type mice. The CCl4 treatment significantly induced genes related to the fibrosis formation in wild-type mice, whereas this was lower in MHCII(Δ/Δ) mice. The liver histology, however, showed no detectable difference between groups, suggesting that the MHC II pathway is not required for the development of hepatic fibrosis induced by CCl4.
Figures



References
-
- Mitchison NA. T-cell-B-cell cooperation. Nature Reviews Immunology. 2004;4:308–312. - PubMed
-
- Singer JB, Lewitzky S, Leroy E, et al. A genome-wide study identifies HLA alleles associated with lumiracoxib-related liver injury. Nature Genetics. 2010;42(8):711–714. - PubMed
-
- Laine L, White WB, Rostom A, Hochberg M. COX-2 selective inhibitors in the treatment of osteoarthritis. Seminars in Arthritis and Rheumatism. 2008;38(3):165–187. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

