Optimizing donor selection for public cord blood banking: influence of maternal, infant, and collection characteristics on cord blood unit quality
- PMID: 23711284
- PMCID: PMC3766489
- DOI: 10.1111/trf.12257
Optimizing donor selection for public cord blood banking: influence of maternal, infant, and collection characteristics on cord blood unit quality
Abstract
Background: Banked unrelated donor umbilical cord blood (CB) has improved access to hematopoietic stem cell transplantation for patients without a suitably matched donor. In a resource-limited environment, ensuring that the public inventory is enriched with high-quality cord blood units (CBUs) addressing the needs of a diverse group of patients is a priority. Identification of donor characteristics correlating with higher CBU quality could guide operational strategies to increase the yield of banked high-quality CBUs.
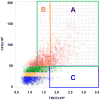
Study design and methods: Characteristics of 5267 CBUs donated to the Carolinas Cord Blood Bank, a public bank participating in the National Cord Blood Inventory, were retrospectively analyzed. Eligible CBUs, collected by trained personnel, were processed using standard procedures. Routine quality and potency metrics (postprocessing total nucleated cell count [post-TNCC], CD34+, colony-forming units [CFUs]) were correlated with maternal, infant, and collection characteristics.
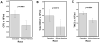
Results: High-quality CBUs were defined as those with higher post-TNCC (>1.25 × 10(9)) with CD34+ and CFUs in the upper quartile. Factors associated with higher CD34+ or CFU content included a shorter interval from collection to processing (<10 hr), younger gestational age (34-37 weeks; CD34+ and CFUs), Caucasian race, higher birthweight (>3500 g), and larger collection volumes (>80 mL).
Conclusions: We describe characteristics identifying high-quality CBUs, which can be used to inform strategies for CBU collection for public banks. Efforts should be made to prioritize collections from larger babies born before 38 weeks of gestation. CBUs should be rapidly transported to the processing laboratory. The lower quality of CBUs from non-Caucasian donors highlights the challenges of building a racially diverse public CB inventory.
© 2013 American Association of Blood Banks.
Conflict of interest statement
Conflict of Interest: The authors declare that they have no conflicts of interest relevant to the manuscript submitted to TRANSFUSION.
Figures
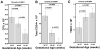


Comment in
-
Increase the quality of banked cord blood units without limiting HLA diversity: how cord blood banks could face this dilemma.Transfusion. 2014 Feb;54(2):495-6. doi: 10.1111/trf.12439. Transfusion. 2014. PMID: 24517133 No abstract available.
-
In reply.Transfusion. 2014 Feb;54(2):496. doi: 10.1111/trf.12465. Transfusion. 2014. PMID: 24517135 No abstract available.
Similar articles
-
Influence of maternal, infant, and collection characteristics on high-quality cord blood units in Guangzhou Cord Blood Bank.Transfusion. 2015 Sep;55(9):2158-67. doi: 10.1111/trf.13126. Epub 2015 May 21. Transfusion. 2015. PMID: 25994982
-
Predictors of cord blood unit cell content in a volume unrestricted large series collections: a chance for a fast and cheap multiparameter selection model.Stem Cell Res Ther. 2022 Jun 11;13(1):246. doi: 10.1186/s13287-022-02915-y. Stem Cell Res Ther. 2022. PMID: 35690786 Free PMC article.
-
Characterization of banked umbilical cord blood hematopoietic progenitor cells and lymphocyte subsets and correlation with ethnicity, birth weight, sex, and type of delivery: a Cord Blood Transplantation (COBLT) Study report.Transfusion. 2005 Jun;45(6):856-66. doi: 10.1111/j.1537-2995.2005.04429.x. Transfusion. 2005. PMID: 15934982
-
Umbilical cord blood quality and quantity: Collection up to transplantation.Asian J Transfus Sci. 2019 Jul-Dec;13(2):79-89. doi: 10.4103/ajts.AJTS_124_18. Epub 2019 Dec 3. Asian J Transfus Sci. 2019. PMID: 31896912 Free PMC article. Review.
-
Human umbilical cord blood banking and transplantation: a state of the art.Eur J Obstet Gynecol Reprod Biol. 2000 May;90(1):13-25. doi: 10.1016/s0301-2115(99)00214-6. Eur J Obstet Gynecol Reprod Biol. 2000. PMID: 10767505 Review.
Cited by
-
Detecting primitive hematopoietic stem cells in total nucleated and mononuclear cell fractions from umbilical cord blood segments and units.J Transl Med. 2015;13:94. doi: 10.1186/s12967-015-0434-z. Epub 2015 Mar 18. J Transl Med. 2015. PMID: 25784613 Free PMC article.
-
Selection Criteria of Cord Blood Units for Platelet Gel Production: Proposed Directions from Hellenic Cord Blood Bank. Comment on Mallis et al. Short Term Results of Fibrin Gel Obtained from Cord Blood Units: A Preliminary in Vitro Study. Bioengineering 2019, 6, 66.Bioengineering (Basel). 2021 Apr 27;8(5):53. doi: 10.3390/bioengineering8050053. Bioengineering (Basel). 2021. PMID: 33925496 Free PMC article.
-
Unrelated cord blood transplantation for adult patients with acute myeloid leukemia: higher incidence of acute graft-versus-host disease and lower survival in male patients transplanted with female unrelated cord blood--a report from Eurocord, the Acute Leukemia Working Party, and the Cord Blood Committee of the Cellular Therapy and Immunobiology Working Party of the European Group for Blood and Marrow Transplantation.J Hematol Oncol. 2015 Oct 6;8:107. doi: 10.1186/s13045-015-0207-4. J Hematol Oncol. 2015. PMID: 26445106 Free PMC article.
-
Hematopoietic stem and progenitor cell harvesting: technical advances and clinical utility.J Blood Med. 2015 Feb 18;6:55-67. doi: 10.2147/JBM.S52783. eCollection 2015. J Blood Med. 2015. PMID: 25733943 Free PMC article. Review.
-
Cord blood collection and banking from a population with highly diverse geographic origins increase HLA diversity in the registry and do not lower the proportion of validated cord blood units: experience of the Marseille Cord Blood Bank.Bone Marrow Transplant. 2015 Apr;50(4):531-5. doi: 10.1038/bmt.2014.314. Epub 2015 Jan 26. Bone Marrow Transplant. 2015. PMID: 25621799
References
-
- Kurtzberg J, Laughlin M, Graham ML, Smith C, Olson JF, Halperin EC, Ciocci G, Carrier C, Stevens CE, Rubinstein P. Placental Blood as a Source of Hematopoietic Stem Cells for Transplantation into Unrelated Recipients. N Engl J Med. 1996;335:157–66. - PubMed
-
- Kurtzberg J, Prasad VK, Carter SL, Wagner JE, Baxter-Lowe LA, Wall D, Kapoor N, Guinan EC, Feig SA, Wagner EL, Kernan NA on behalf of the CSC. Results of the Cord Blood Transplantation Study (COBLT): clinical outcomes of unrelated donor umbilical cord blood transplantation in pediatric patients with hematologic malignancies. Blood. 2008;112:4318–27. - PMC - PubMed
-
- Eapen M, Rubinstein P, Zhang M-J, Stevens C, Kurtzberg J, Scaradavou A, Loberiza FR, Champlin RE, Klein JP, Horowitz MM, Wagner JE. Outcomes of transplantation of unrelated donor umbilical cord blood and bone marrow in children with acute leukaemia: a comparison study. Lancet. 2007;369:1947–54. - PubMed
-
- Kurtzberg J, Cairo MS, Fraser JK, Baxter-Lowe L, Cohen G, Carter SL, Kernan NA. Results of the cord blood transplantation (COBLT) study unrelated donor banking program. Transfusion. 2005;45:842–55. - PubMed
-
- Rubinstein P, Carrier C, Scaradavou A, Kurtzberg J, Adamson J, Migliaccio AR, Berkowitz RL, Cabbad M, Dobrila NL, Taylor PE, Rosenfield RE, Stevens CE. Outcomes among 562 Recipients of Placental-Blood Transplants from Unrelated Donors. N Engl J Med. 1998;339:1565–77. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

