Depletion of stromal cells expressing fibroblast activation protein-α from skeletal muscle and bone marrow results in cachexia and anemia
- PMID: 23712428
- PMCID: PMC3674708
- DOI: 10.1084/jem.20122344
Depletion of stromal cells expressing fibroblast activation protein-α from skeletal muscle and bone marrow results in cachexia and anemia
Abstract
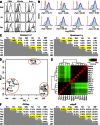
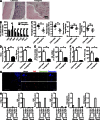
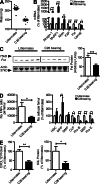
Fibroblast activation protein-α (FAP) identifies stromal cells of mesenchymal origin in human cancers and chronic inflammatory lesions. In mouse models of cancer, they have been shown to be immune suppressive, but studies of their occurrence and function in normal tissues have been limited. With a transgenic mouse line permitting the bioluminescent imaging of FAP(+) cells, we find that they reside in most tissues of the adult mouse. FAP(+) cells from three sites, skeletal muscle, adipose tissue, and pancreas, have highly similar transcriptomes, suggesting a shared lineage. FAP(+) cells of skeletal muscle are the major local source of follistatin, and in bone marrow they express Cxcl12 and KitL. Experimental ablation of these cells causes loss of muscle mass and a reduction of B-lymphopoiesis and erythropoiesis, revealing their essential functions in maintaining normal muscle mass and hematopoiesis, respectively. Remarkably, these cells are altered at these sites in transplantable and spontaneous mouse models of cancer-induced cachexia and anemia. Thus, the FAP(+) stromal cell may have roles in two adverse consequences of cancer: their acquisition by tumors may cause failure of immunosurveillance, and their alteration in normal tissues contributes to the paraneoplastic syndromes of cachexia and anemia.
Figures




References
-
- Acharyya S., Butchbach M.E., Sahenk Z., Wang H., Saji M., Carathers M., Ringel M.D., Skipworth R.J., Fearon K.C., Hollingsworth M.A., et al. 2005. Dystrophin glycoprotein complex dysfunction: a regulatory link between muscular dystrophy and cancer cachexia. Cancer Cell. 8:421–432 10.1016/j.ccr.2005.10.004 - DOI - PubMed
-
- Aertgeerts K., Levin I., Shi L., Snell G.P., Jennings A., Prasad G.S., Zhang Y., Kraus M.L., Salakian S., Sridhar V., et al. 2005. Structural and kinetic analysis of the substrate specificity of human fibroblast activation protein alpha. J. Biol. Chem. 280:19441–19444 10.1074/jbc.C500092200 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

