The bioelectrical basis and validity of gastrointestinal extracellular slow wave recordings
- PMID: 23713030
- PMCID: PMC3784199
- DOI: 10.1113/jphysiol.2013.254292
The bioelectrical basis and validity of gastrointestinal extracellular slow wave recordings
Abstract
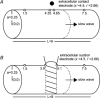
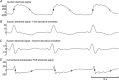
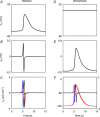
Gastrointestinal extracellular recordings have been a core technique in motility research for a century. However, the bioelectrical basis of extracellular data has recently been challenged by claims that these techniques preferentially assay movement artifacts, cannot reproduce the underlying slow wave kinetics, and misrepresent the true slow wave frequency. These claims motivated this joint experimental-theoretical study, which aimed to define the sources and validity of extracellular potentials. In vivo extracellular recordings and video capture were performed in the porcine jejunum, before and after intra-arterial nifedipine administration. Gastric extracellular recordings were recorded simultaneously using conventional serosal contact and suction electrodes, and biphasic and monophasic extracellular potentials were simulated in a biophysical model. Contractions were abolished by nifedipine, but extracellular slow waves persisted, with unchanged amplitude, downstroke rate, velocity, and downstroke width (P>0.10 for all), at reduced frequency (24% lower; P=0.03). Simultaneous suction and conventional serosal extracellular recordings were identical in phase (frequency and activation-recovery interval), but varied in morphology (monophasic vs. biphasic; downstroke rate and amplitude: P<0.0001). Simulations demonstrated the field contribution of current flow to extracellular potential and quantified the effects of localised depolarisation due to suction pressure on extracellular potential morphology. In sum, these results demonstrate that gastrointestinal extracellular slow wave recordings cannot be explained by motion artifacts, and are of a bioelectrical origin that is highly consistent with the underlying biophysics of slow wave propagation. Motion suppression is shown to be unnecessary as a routine control in in vivo extracellular studies, supporting the validity of the extant gastrointestinal extracellular literature.
Figures


Comment in
-
The gold standard for interpretation of slow wave frequency in in vitro and in vivo recordings by extracellular electrodes.J Physiol. 2013 Sep 15;591(18):4373-4. doi: 10.1113/jphysiol.2013.260976. J Physiol. 2013. PMID: 24037134 Free PMC article. No abstract available.
References
-
- Alvarez WC, Hosoi K. What has happened to the unobstructed bowel that fails to transport fluids & gas. Am J Surg. 1929;6:569–578.
-
- Alvarez WC, Mahoney LJ. Action currents in stomach and intestine. Am J Physiol Gastrointest Liver Physiol. 1922;58:G476–G493.
-
- Barr RC. Basic Electrophysiology. In: Bronzino JD, editor. The Biomedical Engineering Handbook: Second Edition. Boca Raton, FL, USA: CRC Press LLC; 2000.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

