Image-guided therapies for myocardial repair: concepts and practical implementation
- PMID: 23720377
- PMCID: PMC3859252
- DOI: 10.1093/ehjci/jet038
Image-guided therapies for myocardial repair: concepts and practical implementation
Abstract
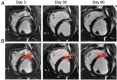
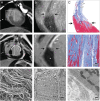
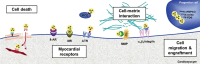
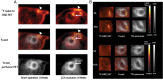
Cell- and molecule-based therapeutic strategies to support wound healing and regeneration after myocardial infarction (MI) are under development. These emerging therapies aim at sustained preservation of ventricular function by enhancing tissue repair after myocardial ischaemia and reperfusion. Such therapies will benefit from guidance with regard to timing, regional targeting, suitable candidate selection, and effectiveness monitoring. Such guidance is effectively obtained by non-invasive tomographic imaging. Infarct size, tissue characteristics, muscle mass, and chamber geometry can be determined by magnetic resonance imaging and computed tomography. Radionuclide imaging can be used for the tracking of therapeutic agents and for the interrogation of molecular mechanisms such as inflammation, angiogenesis, and extracellular matrix activation. This review article portrays the hypothesis that an integrated approach with an early implementation of structural and molecular tomographic imaging in the development of novel therapies will provide a framework for achieving the goal of improved tissue repair after MI.
Keywords: Acute myocardial infarction; Computed tomography; Magnetic resonance; Molecular imaging; Myocardial regeneration; Wound healing.
Figures

Similar articles
-
Cardiovascular magnetic resonance imaging in delivering and evaluating the efficacy of hepatocyte growth factor gene in chronic infarct scar.Cardiovasc Revasc Med. 2011 Mar-Apr;12(2):111-22. doi: 10.1016/j.carrev.2010.05.003. Epub 2010 Oct 20. Cardiovasc Revasc Med. 2011. PMID: 21421190 Free PMC article.
-
Targeted transendocardial therapeutic delivery guided by MRI-x-ray image fusion.Catheter Cardiovasc Interv. 2011 Sep 1;78(3):468-78. doi: 10.1002/ccd.22901. Epub 2011 Mar 16. Catheter Cardiovasc Interv. 2011. PMID: 21413125
-
Acute myocardial infarction: serial cardiac MR imaging shows a decrease in delayed enhancement of the myocardium during the 1st week after reperfusion.Radiology. 2010 Jan;254(1):88-97. doi: 10.1148/radiol.09090660. Radiology. 2010. PMID: 20032144
-
Imaging the healing murine myocardial infarct in vivo: ultrasound, magnetic resonance imaging and fluorescence molecular tomography.Exp Physiol. 2013 Mar;98(3):606-13. doi: 10.1113/expphysiol.2012.064741. Epub 2012 Oct 12. Exp Physiol. 2013. PMID: 23064510 Review.
-
Approaches to Improving Cardiac Structure and Function During and After an Acute Myocardial Infarction: Acute and Chronic Phases.J Cardiovasc Pharmacol Ther. 2016 Jul;21(4):363-7. doi: 10.1177/1074248415616187. Epub 2015 Nov 25. J Cardiovasc Pharmacol Ther. 2016. PMID: 26612091 Review.
Cited by
-
PET imaging of cardiac wound healing using a novel [68Ga]-labeled NGR probe in rat myocardial infarction.Mol Imaging Biol. 2015 Feb;17(1):76-86. doi: 10.1007/s11307-014-0751-2. Epub 2014 Jul 11. Mol Imaging Biol. 2015. PMID: 25011975
-
PET-based myocardial efficiency: Powerful yet under-utilized-now simpler than ever.J Nucl Cardiol. 2018 Dec;25(6):1945-1947. doi: 10.1007/s12350-018-1400-y. Epub 2018 Aug 10. J Nucl Cardiol. 2018. PMID: 30097851 No abstract available.
-
Emerging imaging targets for infiltrative cardiomyopathy: Inflammation and fibrosis.J Nucl Cardiol. 2019 Feb;26(1):208-216. doi: 10.1007/s12350-018-1356-y. Epub 2018 Jul 2. J Nucl Cardiol. 2019. PMID: 29968156 Review.
-
The complex role of cardiovascular imaging in viability testing.Prog Cardiovasc Dis. 2025 Jan-Feb;88:113-125. doi: 10.1016/j.pcad.2024.12.008. Epub 2025 Jan 7. Prog Cardiovasc Dis. 2025. PMID: 39788340 Review.
-
Multimodality Imaging of Inflammation and Ventricular Remodeling in Pressure-Overload Heart Failure.J Nucl Med. 2020 Apr;61(4):590-596. doi: 10.2967/jnumed.119.232488. Epub 2019 Oct 25. J Nucl Med. 2020. PMID: 31653713 Free PMC article.
References
-
- Nahrendorf M, Pittet MJ, Swirski FK. Monocytes: protagonists of infarct inflammation and repair after myocardial infarction. Circulation. 2010;121:2437–45. doi:10.1161/CIRCULATIONAHA.109.916346. - DOI - PMC - PubMed
-
- Frangogiannis NG. Regulation of the inflammatory response in cardiac repair. Circ Res. 2012;110:159–73. doi:10.1161/CIRCRESAHA.111.243162. - DOI - PMC - PubMed
-
- van den Borne SW, Diez J, Blankesteijn WM, Verjans J, Hofstra L, Narula J. Myocardial remodeling after infarction: the role of myofibroblasts. Nat Rev Cardiol. 2010;7:30–7. doi:10.1038/nrcardio.2009.199. - DOI - PubMed
-
- Kempf T, Zarbock A, Vestweber D, Wollert KC. Anti-inflammatory mechanisms and therapeutic opportunities in myocardial infarct healing. J Mol Med (Berl) 2012;90:361–9. doi:10.1007/s00109-011-0847-y. - DOI - PubMed
-
- Dobaczewski M, Gonzalez-Quesada C, Frangogiannis NG. The extracellular matrix as a modulator of the inflammatory and reparative response following myocardial infarction. J Mol Cell Cardiol. 2010;48:504–11. doi:10.1016/j.yjmcc.2009.07.015. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

