Antibody and antiretroviral preexposure prophylaxis prevent cervicovaginal HIV-1 infection in a transgenic mouse model
- PMID: 23720722
- PMCID: PMC3719827
- DOI: 10.1128/JVI.00868-13
Antibody and antiretroviral preexposure prophylaxis prevent cervicovaginal HIV-1 infection in a transgenic mouse model
Abstract
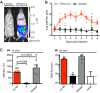
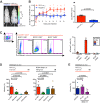
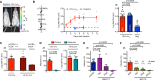
The development of an effective vaccine preventing HIV-1 infection remains elusive. Thus, the development of novel approaches capable of preventing HIV-1 transmission is of paramount importance. However, this is partly hindered by the lack of an easily accessible small-animal model to rapidly measure viral entry. Here, we report the generation of a human CD4- and human CCR5-expressing transgenic luciferase reporter mouse that facilitates measurement of peritoneal and genitomucosal HIV-1 pseudovirus entry in vivo. We show that antibodies and antiretrovirals mediate preexposure protection in this mouse model and that the serum antibody concentration required for protection from cervicovaginal infection is comparable to that required to protect macaques. Our results suggest that this system represents a model for the preclinical evaluation of prophylactic or vaccine candidates. It further supports the idea that broadly neutralizing antibodies should be evaluated for use as preexposure prophylaxis in clinical trials.
Figures
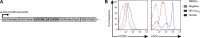

References
-
- Centers for Disease Control 1981. Pneumocystis pneumonia—Los Angeles. MMWR Morb. Mortal. Wkly. Rep. 30:250–252 - PubMed
-
- UNAIDS 2012. Global report: UNAIDS report on the global AIDS epidemic 2012. UNAIDS, Geneva, Switzerland: http://www.unaids.org/en/resources/campaigns/20121120_globalreport2012/g...
-
- Haynes BF, Gilbert PB, McElrath MJ, Zolla-Pazner S, Tomaras GD, Alam SM, Evans DT, Montefiori DC, Karnasuta C, Sutthent R, Liao HX, DeVico AL, Lewis GK, Williams C, Pinter A, Fong Y, Janes H, DeCamp A, Huang Y, Rao M, Billings E, Karasavvas N, Robb ML, Ngauy V, de Souza MS, Paris R, Ferrari G, Bailer RT, Soderberg KA, Andrews C, Berman PW, Frahm N, De Rosa SC, Alpert MD, Yates NL, Shen X, Koup RA, Pitisuttithum P, Kaewkungwal J, Nitayaphan S, Rerks-Ngarm S, Michael NL, Kim JH. 2012. Immune-correlates analysis of an HIV-1 vaccine efficacy trial. N. Engl. J. Med. 366:1275–1286 - PMC - PubMed
-
- Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, Paris R, Premsri N, Namwat C, de Souza M, Adams E, Benenson M, Gurunathan S, Tartaglia J, McNeil JG, Francis DP, Stablein D, Birx DL, Chunsuttiwat S, Khamboonruang C, Thongcharoen P, Robb ML, Michael NL, Kunasol P, Kim JH. 2009. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N. Engl. J. Med. 361:2209–2220 - PubMed
-
- Cohen MS, Chen YQ, McCauley M, Gamble T, Hosseinipour MC, Kumarasamy N, Hakim JG, Kumwenda J, Grinsztejn B, Pilotto JH, Godbole SV, Mehendale S, Chariyalertsak S, Santos BR, Mayer KH, Hoffman IF, Eshleman SH, Piwowar-Manning E, Wang L, Makhema J, Mills LA, de Bruyn G, Sanne I, Eron J, Gallant J, Havlir D, Swindells S, Ribaudo H, Elharrar V, Burns D, Taha TE, Nielsen-Saines K, Celentano D, Essex M, Fleming TR. 2011. Prevention of HIV-1 infection with early antiretroviral therapy. N. Engl. J. Med. 365:493–505 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

