Molecular mechanisms, thermodynamics, and dissociation kinetics of knob-hole interactions in fibrin
- PMID: 23720752
- PMCID: PMC3829353
- DOI: 10.1074/jbc.M113.472365
Molecular mechanisms, thermodynamics, and dissociation kinetics of knob-hole interactions in fibrin
Abstract
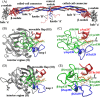
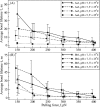
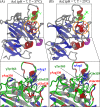
Polymerization of fibrin, the primary structural protein of blood clots and thrombi, occurs through binding of knobs 'A' and 'B' in the central nodule of fibrin monomer to complementary holes 'a' and 'b' in the γ- and β-nodules, respectively, of another monomer. We characterized the A:a and B:b knob-hole interactions under varying solution conditions using molecular dynamics simulations of the structural models of fibrin(ogen) fragment D complexed with synthetic peptides GPRP (knob 'A' mimetic) and GHRP (knob 'B' mimetic). The strength of A:a and B:b knob-hole complexes was roughly equal, decreasing with pulling force; however, the dissociation kinetics were sensitive to variations in acidity (pH 5-7) and temperature (T = 25-37 °C). There were similar structural changes in holes 'a' and 'b' during forced dissociation of the knob-hole complexes: elongation of loop I, stretching of the interior region, and translocation of the moveable flap. The disruption of the knob-hole interactions was not an "all-or-none" transition as it occurred through distinct two-step or single step pathways with or without intermediate states. The knob-hole bonds were stronger, tighter, and more brittle at pH 7 than at pH 5. The B:b knob-hole bonds were weaker, looser, and more compliant than the A:a knob-hole bonds at pH 7 but stronger, tighter, and less compliant at pH 5. Surprisingly, the knob-hole bonds were stronger, not weaker, at elevated temperature (T = 37 °C) compared with T = 25 °C due to the helix-to-coil transition in loop I that helps stabilize the bonds. These results provide detailed qualitative and quantitative characteristics underlying the most significant non-covalent interactions involved in fibrin polymerization.
Keywords: A:a and B:b Knob-Hole Bonds; Biophysics; Dissociation Kinetics; Fibrin; Fibrinogen; Free Energy Landscape; Molecular Dynamics; Protein-Protein Interactions.
Figures



References
-
- Weisel J. W. (2004) The mechanical properties of fibrin for basic scientists and clinicians. Biophys. Chem. 112, 267–276 - PubMed
-
- Ferry J. D. (1988) in Biological and synthetic polymer networks (Kramer O., ed) pp. 41–55, Elsevier, Amsterdam
-
- Weisel J. W. (2008) Enigmas of blood clot elasticity. Science 320, 456–457 - PubMed
-
- Weisel J. W. (2005) Fibrinogen and fibrin. Adv. Protein Chem. 70, 247–299 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

