Immune cells control skin lymphatic electrolyte homeostasis and blood pressure
- PMID: 23722907
- PMCID: PMC3696542
- DOI: 10.1172/JCI60113
Immune cells control skin lymphatic electrolyte homeostasis and blood pressure
Abstract
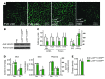
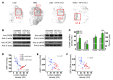
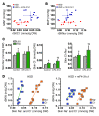
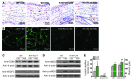
The skin interstitium sequesters excess Na+ and Cl- in salt-sensitive hypertension. Mononuclear phagocyte system (MPS) cells are recruited to the skin, sense the hypertonic electrolyte accumulation in skin, and activate the tonicity-responsive enhancer-binding protein (TONEBP, also known as NFAT5) to initiate expression and secretion of VEGFC, which enhances electrolyte clearance via cutaneous lymph vessels and increases eNOS expression in blood vessels. It is unclear whether this local MPS response to osmotic stress is important to systemic blood pressure control. Herein, we show that deletion of TonEBP in mouse MPS cells prevents the VEGFC response to a high-salt diet (HSD) and increases blood pressure. Additionally, an antibody that blocks the lymph-endothelial VEGFC receptor, VEGFR3, selectively inhibited MPS-driven increases in cutaneous lymphatic capillary density, led to skin Cl- accumulation, and induced salt-sensitive hypertension. Mice overexpressing soluble VEGFR3 in epidermal keratinocytes exhibited hypoplastic cutaneous lymph capillaries and increased Na+, Cl-, and water retention in skin and salt-sensitive hypertension. Further, we found that HSD elevated skin osmolality above plasma levels. These results suggest that the skin contains a hypertonic interstitial fluid compartment in which MPS cells exert homeostatic and blood pressure-regulatory control by local organization of interstitial electrolyte clearance via TONEBP and VEGFC/VEGFR3-mediated modification of cutaneous lymphatic capillary function.
Figures

References
-
- Guyton AC, Coleman TC, Cowley AW, Jr, Manning RD, Jr, Norman RA, Jr, Ferguson JD. A systems analysis approach to understanding long-range arterial blood pressure control and hypertension. Circ Res. 1974;35:159–176. doi: 10.1161/01.RES.35.2.159. - DOI
-
- Haljamae H. Anatomy of the interstitial tissue. Lymphology. 1978;11(4):128–132. - PubMed
-
- Haljamae H, Linde A, Amundson B. Comparative analyses of capsular fluid and interstitial fluid. Am J Physiol. 1974;227(5):1199–1205. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

