Fasting induces nuclear factor E2-related factor 2 and ATP-binding Cassette transporters via protein kinase A and Sirtuin-1 in mouse and human
- PMID: 23725046
- PMCID: PMC3880903
- DOI: 10.1089/ars.2012.5082
Fasting induces nuclear factor E2-related factor 2 and ATP-binding Cassette transporters via protein kinase A and Sirtuin-1 in mouse and human
Abstract
Aims: The purpose of this study was to determine whether 3'-5'-cyclic adenosine monophosphate (cAMP)-protein kinase A (PKA) and Sirtuin-1 (SIRT1) dependent mechanisms modulate ATP-binding Cassette (ABC) transport protein expression. ABC transport proteins (ABCC2-4) are essential for chemical elimination from hepatocytes and biliary excretion. Nuclear factor-E2 related-factor 2 (NRF2) is a transcription factor that mediates ABCC induction in response to chemical inducers and liver injury. However, a role for NRF2 in the regulation of transporter expression in nonchemical models of liver perturbation is largely undescribed.
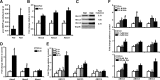
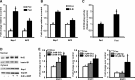
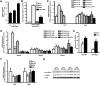
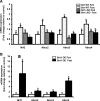
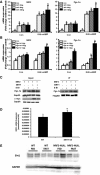
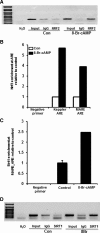
Results: Here we show that fasting increased NRF2 target gene expression through NRF2- and SIRT1-dependent mechanisms. In intact mouse liver, fasting induces NRF2 target gene expression by at least 1.5 to 5-fold. In mouse and human hepatocytes, treatment with 8-Bromoadenosine-cAMP, a cAMP analogue, increased NRF2 target gene expression and antioxidant response element activity, which was decreased by the PKA inhibitor, H-89. Moreover, fasting induced NRF2 target gene expression was decreased in liver and hepatocytes of SIRT1 liver-specific null mice and NRF2-null mice. Lastly, NRF2 and SIRT1 were recruited to MAREs and Antioxidant Response Elements (AREs) in the human ABCC2 promoter.
Innovation: Oxidative stress mediated NRF2 activation is well described, yet the influence of basic metabolic processes on NRF2 activation is just emerging.
Conclusion: The current data point toward a novel role of nutrient status in regulation of NRF2 activity and the antioxidant response, and indicates that cAMP/PKA and SIRT1 are upstream regulators for fasting-induced activation of the NRF2-ARE pathway.
Figures

Similar articles
-
Oxidative and electrophilic stress induces multidrug resistance-associated protein transporters via the nuclear factor-E2-related factor-2 transcriptional pathway.Hepatology. 2007 Nov;46(5):1597-610. doi: 10.1002/hep.21831. Hepatology. 2007. PMID: 17668877
-
Nicotinamide ameliorates palmitate-induced ER stress in hepatocytes via cAMP/PKA/CREB pathway-dependent Sirt1 upregulation.Biochim Biophys Acta. 2015 Nov;1853(11 Pt A):2929-36. doi: 10.1016/j.bbamcr.2015.09.003. Epub 2015 Sep 6. Biochim Biophys Acta. 2015. PMID: 26352206 Free PMC article.
-
Constitutive activation of nuclear factor-E2-related factor 2 induces biotransformation enzyme and transporter expression in livers of mice with hepatocyte-specific deletion of Kelch-like ECH-associated protein 1.J Biochem Mol Toxicol. 2011 Sep-Oct;25(5):320-9. doi: 10.1002/jbt.20392. Epub 2011 May 2. J Biochem Mol Toxicol. 2011. PMID: 21538727 Free PMC article.
-
The Nrf2-antioxidant response element pathway: a target for regulating energy metabolism.J Nutr Biochem. 2012 Oct;23(10):1201-6. doi: 10.1016/j.jnutbio.2012.03.005. Epub 2012 Jul 21. J Nutr Biochem. 2012. PMID: 22819548 Review.
-
Role of Nrf2 in cancer photodynamic therapy: regulation of human ABC transporter ABCG2.J Pharm Sci. 2013 Sep;102(9):3058-69. doi: 10.1002/jps.23563. Epub 2013 May 6. J Pharm Sci. 2013. PMID: 23650051 Review.
Cited by
-
Biomimetics - Nature's roadmap to insights and solutions for burden of lifestyle diseases.J Intern Med. 2020 Mar;287(3):238-251. doi: 10.1111/joim.12982. Epub 2019 Oct 22. J Intern Med. 2020. PMID: 31639885 Free PMC article. Review.
-
Deficiency in Nrf2 transcription factor decreases adipose tissue mass and hepatic lipid accumulation in leptin-deficient mice.Obesity (Silver Spring). 2015 Feb;23(2):335-44. doi: 10.1002/oby.20929. Epub 2014 Nov 29. Obesity (Silver Spring). 2015. PMID: 25451536 Free PMC article.
-
Isoliquiritigenin prevents hyperglycemia-induced renal injuries by inhibiting inflammation and oxidative stress via SIRT1-dependent mechanism.Cell Death Dis. 2020 Dec 7;11(12):1040. doi: 10.1038/s41419-020-03260-9. Cell Death Dis. 2020. PMID: 33288747 Free PMC article.
-
Matrine promotes mitochondrial biosynthesis and reduces oxidative stress in experimental optic neuritis.Front Pharmacol. 2022 Sep 27;13:936632. doi: 10.3389/fphar.2022.936632. eCollection 2022. Front Pharmacol. 2022. PMID: 36238552 Free PMC article.
-
NRF2 maintains redox balance via ME1 and NRF2 inhibitor synergizes with venetoclax in NPM1-mutated acute myeloid leukemia.Cancer Metab. 2025 Jun 18;13(1):32. doi: 10.1186/s40170-025-00401-6. Cancer Metab. 2025. PMID: 40533864 Free PMC article.
References
-
- Aleksunes LM, Scheffer GL, Jakowski AB, Pruimboom-Brees IM, and Manautou JE. Coordinated expression of multidrug resistance-associated proteins (Mrps) in mouse liver during toxicant-induced injury. Toxicol Sci 89: 370–379, 2006 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

