Lysosome-related organelles: unusual compartments become mainstream
- PMID: 23726022
- PMCID: PMC3729921
- DOI: 10.1016/j.ceb.2013.04.008
Lysosome-related organelles: unusual compartments become mainstream
Abstract
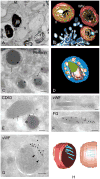
Lysosome-related organelles (LROs) comprise a group of cell type-specific subcellular compartments with unique composition, morphology and structure that share some features with endosomes and lysosomes and that function in varied processes such as pigmentation, hemostasis, lung plasticity and immunity. In recent years, studies of genetic diseases in which LRO functions are compromised have provided new insights into the mechanisms of LRO biogenesis and the regulated secretion of LRO contents. These insights have revealed previously unappreciated specialized endosomal sorting processes in all cell types, and are expanding our views of the plasticity of the endosomal and secretory systems in adapting to cell type-specific needs.
Copyright © 2013 Elsevier Ltd. All rights reserved.
Figures


References
-
- Michaux G, Abbitt KB, Collinson LM, Haberichter SL, Norman KE, Cutler DF. The physiological function of von Willebrand’s factor depends on its tubular storage in endothelial Weibel-Palade bodies. Dev Cell. 2006;10:223–232. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

