Reduced SNAP-25 alters short-term plasticity at developing glutamatergic synapses
- PMID: 23732542
- PMCID: PMC3701242
- DOI: 10.1038/embor.2013.75
Reduced SNAP-25 alters short-term plasticity at developing glutamatergic synapses
Abstract
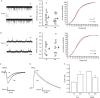
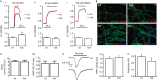
SNAP-25 is a key component of the synaptic-vesicle fusion machinery, involved in several psychiatric diseases including schizophrenia and ADHD. SNAP-25 protein expression is lower in different brain areas of schizophrenic patients and in ADHD mouse models. How the reduced expression of SNAP-25 alters the properties of synaptic transmission, leading to a pathological phenotype, is unknown. We show that, unexpectedly, halved SNAP-25 levels at 13-14 DIV not only fail to impair synaptic transmission but instead enhance evoked glutamatergic neurotransmission. This effect is possibly dependent on presynaptic voltage-gated calcium channel activity and is not accompanied by changes in spontaneous quantal events or in the pool of readily releasable synaptic vesicles. Notably, synapses of 13-14 DIV neurons with reduced SNAP-25 expression show paired-pulse depression as opposed to paired-pulse facilitation occurring in their wild-type counterparts. This phenotype disappears with synapse maturation. As alterations in short-term plasticity represent a new mechanism contributing to cognitive impairments in intellectual disabilities, our data provide mechanistic clues for neuronal circuit alterations in psychiatric diseases characterized by reduced expression of SNAP-25.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures


Comment in
-
A dual role of SNAP-25 as carrier and guardian of synaptic transmission.EMBO Rep. 2013 Jul;14(7):579-80. doi: 10.1038/embor.2013.74. Epub 2013 Jun 4. EMBO Rep. 2013. PMID: 23732543 Free PMC article. No abstract available.
References
-
- Jahn RSR (2006) SNAREs—engines for membrane fusion. Nat Rev Mol Cell Biol 7: 631–643 - PubMed
-
- Washbourne P et al. (2002) Genetic ablation of the t-SNARESNAP-25 distinguishes mechanisms of neuroexocytosis. Nat Neurosci 5: 19–26 - PubMed
-
- Catterall WA, Few AP (2008) Calcium channel regulation and presynaptic plasticity. Neuron 59: 882–901 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

