The kallikrein-kinin system as a regulator of cardiovascular and renal function
- PMID: 23737209
- PMCID: PMC4685708
- DOI: 10.1002/cphy.c100053
The kallikrein-kinin system as a regulator of cardiovascular and renal function
Abstract
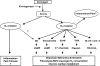
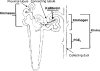
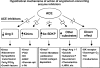
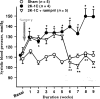
Autocrine, paracrine, endocrine, and neuroendocrine hormonal systems help regulate cardio-vascular and renal function. Any change in the balance among these systems may result in hypertension and target organ damage, whether the cause is genetic, environmental or a combination of the two. Endocrine and neuroendocrine vasopressor hormones such as the renin-angiotensin system (RAS), aldosterone, and catecholamines are important for regulation of blood pressure and pathogenesis of hypertension and target organ damage. While the role of vasodepressor autacoids such as kinins is not as well defined, there is increasing evidence that they are not only critical to blood pressure and renal function but may also oppose remodeling of the cardiovascular system. Here we will primarily be concerned with kinins, which are oligopeptides containing the aminoacid sequence of bradykinin. They are generated from precursors known as kininogens by enzymes such as tissue (glandular) and plasma kallikrein. Some of the effects of kinins are mediated via autacoids such as eicosanoids, nitric oxide (NO), endothelium-derived hyperpolarizing factor (EDHF), and/or tissue plasminogen activator (tPA). Kinins help protect against cardiac ischemia and play an important part in preconditioning as well as the cardiovascular and renal protective effects of angiotensin-converting enzyme (ACE) and angiotensin type 1 receptor blockers (ARB). But the role of kinins in the pathogenesis of hypertension remains controversial. A study of Utah families revealed that a dominant kallikrein gene expressed as high urinary kallikrein excretion was associated with a decreased risk of essential hypertension. Moreover, researchers have identified a restriction fragment length polymorphism (RFLP) that distinguishes the kallikrein gene family found in one strain of spontaneously hypertensive rats (SHR) from a homologous gene in normotensive Brown Norway rats, and in recombinant inbred substrains derived from these SHR and Brown Norway rats this RFLP cosegregated with an increase in blood pressure. However, humans, rats and mice with a deficiency in one or more components of the kallikrein-kinin-system (KKS) or chronic KKS blockade do not have hypertension. In the kidney, kinins are essential for proper regulation of papillary blood flow and water and sodium excretion. B2-KO mice appear to be more sensitive to the hypertensinogenic effect of salt. Kinins are involved in the acute antihypertensive effects of ACE inhibitors but not their chronic effects (save for mineralocorticoid-salt-induced hypertension). Kinins appear to play a role in the pathogenesis of inflammatory diseases such as arthritis and skin inflammation; they act on innate immunity as mediators of inflammation by promoting maturation of dendritic cells, which activate the body's adaptive immune system and thereby stimulate mechanisms that promote inflammation. On the other hand, kinins acting via NO contribute to the vascular protective effect of ACE inhibitors during neointima formation. In myocardial infarction produced by ischemia/reperfusion, kinins help reduce infarct size following preconditioning or treatment with ACE inhibitors. In heart failure secondary to infarction, the therapeutic effects of ACE inhibitors are partially mediated by kinins via release of NO, while drugs that activate the angiotensin type 2 receptor act in part via kinins and NO. Thus kinins play an important role in regulation of cardiovascular and renal function as well as many of the beneficial effects of ACE inhibitors and ARBs on target organ damage in hypertension.
© 2011 American Physiological Society. Compr Physiol 1:699-729, 2011.
Figures



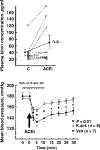


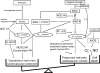
References
-
- Farmer SG, editor. The Kinin System (The Handbook of lmmunopharmacology) San Diego, CA: Academic Press Inc; 1997. pp. 1–323.
-
- Abadir PM, Carey RM, Siragy HM. Angiotensin AT2 receptors directly stimulate renal nitric oxide in bradykinin B2-receptor-null mice. Hypertension. 2003;42:600–604. - PubMed
-
- Abadir PM, Periasamy A, Carey RM, Siragy HM. Angiotensin II type 2 receptor-bradykinin B2 receptor functional heterodimerization. Hypertension. 2006;48:1–7. - PubMed
-
- Abbas A, Gorelik G, Carbini LA, Scicli AG. Angiotensin-(1-7) induces bradykinin-mediated hypotensive responses in anesthetized rats. Hypertension. 1997;30:217–221. - PubMed
-
- AbdAlla S, Abdel-Baset A, Lother H, el Massiery A, Quitterer U. Mesangial AT1/B2 receptor heterodimers contribute to angiotensin II hyperresponsiveness in experimental hypertension. J Mol Neurosci. 2005;26:185–192. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

