Amygdala-dependent fear is regulated by Oprl1 in mice and humans with PTSD
- PMID: 23740899
- PMCID: PMC3732318
- DOI: 10.1126/scitranslmed.3005656
Amygdala-dependent fear is regulated by Oprl1 in mice and humans with PTSD
Abstract
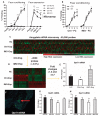
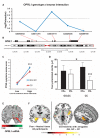
The amygdala-dependent molecular mechanisms driving the onset and persistence of posttraumatic stress disorder (PTSD) are poorly understood. Recent observational studies have suggested that opioid analgesia in the aftermath of trauma may decrease the development of PTSD. Using a mouse model of dysregulated fear, we found altered expression within the amygdala of the Oprl1 gene (opioid receptor-like 1), which encodes the amygdala nociceptin (NOP)/orphanin FQ receptor (NOP-R). Systemic and central amygdala infusion of SR-8993, a new highly selective NOP-R agonist, impaired fear memory consolidation. In humans, a single-nucleotide polymorphism (SNP) within OPRL1 is associated with a self-reported history of childhood trauma and PTSD symptoms (n = 1847) after a traumatic event. This SNP is also associated with physiological startle measures of fear discrimination and magnetic resonance imaging analysis of amygdala-insula functional connectivity. Together, these data suggest that Oprl1 is associated with amygdala function, fear processing, and PTSD symptoms. Further, our data suggest that activation of the Oprl1/NOP receptor may interfere with fear memory consolidation, with implications for prevention of PTSD after a traumatic event.
Figures





Comment in
-
Preventing the pain of PTSD.Sci Transl Med. 2013 Jun 5;5(188):188fs22. doi: 10.1126/scitranslmed.3006635. Sci Transl Med. 2013. PMID: 23740896
References
-
- Phelps EA, LeDoux JE. Contributions of the amygdala to emotion processing: From animal models to human behavior. Neuron. 2005;48:175–187. - PubMed
MeSH terms
Substances
Associated data
- Actions
Grants and funding
- R01 AA017943/AA/NIAAA NIH HHS/United States
- R56 MH071537/MH/NIMH NIH HHS/United States
- R01 MH071537/MH/NIMH NIH HHS/United States
- R01 MH096764/MH/NIMH NIH HHS/United States
- P51 RR000165/RR/NCRR NIH HHS/United States
- U01 AA018665/AA/NIAAA NIH HHS/United States
- UL1 TR000454/TR/NCATS NIH HHS/United States
- R01 DA035055/DA/NIDA NIH HHS/United States
- R21 MH092576/MH/NIMH NIH HHS/United States
- R21 MH098212/MH/NIMH NIH HHS/United States
- R01 DA019624/DA/NIDA NIH HHS/United States
- R01 DA035056/DA/NIDA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

