Dephosphorylation of D-peptide derivatives to form biofunctional, supramolecular nanofibers/hydrogels and their potential applications for intracellular imaging and intratumoral chemotherapy
- PMID: 23742714
- PMCID: PMC3730259
- DOI: 10.1021/ja404215g
Dephosphorylation of D-peptide derivatives to form biofunctional, supramolecular nanofibers/hydrogels and their potential applications for intracellular imaging and intratumoral chemotherapy
Abstract
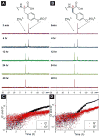
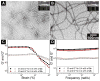
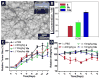
D-Peptides, as the enantiomers of the naturally occurring L-peptides, usually resist endogenous proteases and are presumably insensitive to most enzymes. But, it is unclear whether or how a phosphatase catalyzes the dephosphorylation from D-peptides. In this work, we examine the formation of the nanofibers of D-peptides via enzymatic dephosphorylation. By comparing the enzymatic hydrogelation of L-peptide and D-peptide based hydrogelators, we find that the chirality of the precursors of the hydrogelators affects little on the enzymatic hydrogelation resulted from the removal of the phosphate group from a tyrosine phosphate residue. The attachment of a therapeutic agent (e.g., taxol) or a fluorophore (e.g., 4-nitro-2,1,3-benzoxadiazole) to the D-peptide based hydrogelators affords a new type of biostable or biocompatible hydrogelators, which may find applications in intratumoral chemotherapy or intracellular imaging, respectively. This work, as the first comprehensive and systematic study of the unexpected enzymatic dephosphorylation of D-peptides, illustrates a useful approach to generate supramolecular hydrogels that have both biostability and other desired functions.
Figures



Similar articles
-
Biofunctional supramolecular hydrogels fabricated from a short self-assembling peptide modified with bioactive sequences for the 3D culture of breast cancer MCF-7 cells.Bioorg Med Chem. 2021 Sep 15;46:116345. doi: 10.1016/j.bmc.2021.116345. Epub 2021 Aug 11. Bioorg Med Chem. 2021. PMID: 34416510
-
Introducing D-amino acid or simple glycoside into small peptides to enable supramolecular hydrogelators to resist proteolysis.Langmuir. 2012 Sep 18;28(37):13512-7. doi: 10.1021/la302583a. Epub 2012 Sep 4. Langmuir. 2012. PMID: 22906360 Free PMC article.
-
The conjugates of forky peptides and nonsteroidal anti-inflammatory drugs (NSAID) self-assemble into supramolecular hydrogels for prostate cancer-specific drug delivery.J Mater Chem B. 2019 Jan 21;7(3):469-476. doi: 10.1039/c8tb02307g. Epub 2018 Dec 20. J Mater Chem B. 2019. PMID: 32254734
-
Supramolecular Hydrogelators and Hydrogels: From Soft Matter to Molecular Biomaterials.Chem Rev. 2015 Dec 23;115(24):13165-307. doi: 10.1021/acs.chemrev.5b00299. Epub 2015 Dec 8. Chem Rev. 2015. PMID: 26646318 Free PMC article. Review.
-
Enzyme-Instructed Self-Assembly (EISA) and Hydrogelation of Peptides.Adv Mater. 2020 Jan;32(3):e1805798. doi: 10.1002/adma.201805798. Epub 2019 Apr 24. Adv Mater. 2020. PMID: 31018025 Review.
Cited by
-
Peptide-drug conjugates as effective prodrug strategies for targeted delivery.Adv Drug Deliv Rev. 2017 Feb;110-111:112-126. doi: 10.1016/j.addr.2016.06.015. Epub 2016 Jun 29. Adv Drug Deliv Rev. 2017. PMID: 27370248 Free PMC article. Review.
-
Ligand-Receptor Interaction Modulates the Energy Landscape of Enzyme-Instructed Self-Assembly of Small Molecules.J Am Chem Soc. 2016 Nov 30;138(47):15397-15404. doi: 10.1021/jacs.6b07677. Epub 2016 Nov 15. J Am Chem Soc. 2016. PMID: 27797504 Free PMC article.
-
Directly observing intracellular nanoparticle formation with nanocomputed tomography.Sci Adv. 2020 Oct 23;6(43):eaba3190. doi: 10.1126/sciadv.aba3190. Print 2020 Oct. Sci Adv. 2020. PMID: 33097531 Free PMC article.
-
Self-Assembly, Bioactivity, and Nanomaterials Applications of Peptide Conjugates with Bulky Aromatic Terminal Groups.ACS Appl Bio Mater. 2023 Feb 20;6(2):384-409. doi: 10.1021/acsabm.2c01041. Epub 2023 Feb 3. ACS Appl Bio Mater. 2023. PMID: 36735801 Free PMC article. Review.
-
Stimuli-Responsive Supramolecular Hydrogels and Their Applications in Regenerative Medicine.Macromol Biosci. 2019 Jan;19(1):e1800259. doi: 10.1002/mabi.201800259. Epub 2018 Oct 8. Macromol Biosci. 2019. PMID: 30295012 Free PMC article. Review.
References
-
- Terech P, Weiss RG. Chem Rev. 1997;97:3133–3159. - PubMed
- Heeres A, van der Pol C, Stuart MCA, Friggeri A, Feringa BL, van Esch J. J Am Chem Soc. 2003;125:14252–14253. - PubMed
- Mukhopadhyay S, Maitra U, Ira, Krishnamoorthy G, Schmidt J, Talmon Y. J Am Chem Soc. 2004;126:15905–15914. - PubMed
- Zhang Y, Yang ZM, Yuan F, Gu HW, Gao P, Xu B. J Am Chem Soc. 2004;126:15028–15029. - PubMed
- Vemula PK, Li J, John G. J Am Chem Soc. 2006;128:8932–8938. - PubMed
- Yang ZM, Liang GL, Wang L, Xu B. J Am Chem Soc. 2006;128:3038–3043. - PubMed
- Cai W, Wang GT, Du P, Wang RX, Jiang XK, Li ZT. J Am Chem Soc. 2008;130:13450–13459. - PubMed
- Ma ML, Kuang Y, Gao Y, Zhang Y, Gao P, Xu B. J Am Chem Soc. 2010;132:2719–2728. - PubMed
- Li XM, Kuang Y, Shi JF, Gao Y, Lin HC, Xu B. J Am Chem Soc. 2011;133:17513–17518. - PMC - PubMed
- Zheng WT, Gao J, Song LJ, Chen CY, Guan D, Wang ZH, Li ZB, Kong DL, Yang ZM. J Am Chem Soc. 2013;135:266–271. - PubMed
- Xing BG, Yu CW, Chow KH, Ho PL, Fu DG, Xu B. J Am Chem Soc. 2002;124:14846–14847. - PubMed
-
- Estroff LA, Hamilton AD. Chem Rev. 2004;104:1201–1217. - PubMed
-
- Hartgerink JD, Beniash E, Stupp SI. Science. 2001;294:1684–1688. - PubMed
- Holmes TC, de Lacalle S, Su X, Liu GS, Rich A, Zhang SG. Proc Natl Acad Sci U S A. 2000;97:6728–6733. - PMC - PubMed
- Haines LA, Rajagopal K, Ozbas B, Salick DA, Pochan DJ, Schneider JP. J Am Chem Soc. 2005;127:17025–17029. - PMC - PubMed
- Galler KM, Aulisa L, Regan KR, D’Souza RN, Hartgerink JD. J Am Chem Soc. 2010;132:3217–3223. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

