Endothelial nitric oxide deficiency promotes Alzheimer's disease pathology
- PMID: 23745722
- PMCID: PMC3825764
- DOI: 10.1111/jnc.12334
Endothelial nitric oxide deficiency promotes Alzheimer's disease pathology
Abstract
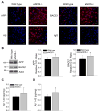
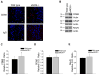
Aging and the presence of cerebrovascular disease are associated with increased incidence of Alzheimer's disease. A common feature of aging and cerebrovascular disease is decreased endothelial nitric oxide (NO). We studied the effect of a loss of endothelium derived NO on amyloid precursor protein (APP) related phenotype in late middle aged (LMA) (14-15 month) endothelial nitric oxide synthase deficient (eNOS(-/-) ) mice. APP, β-site APP cleaving enzyme (BACE) 1, and amyloid beta (Aβ) levels were significantly higher in the brains of LMA eNOS(-/-) mice as compared with LMA wild-type controls. APP and Aβ1-40 were increased in hippocampal tissue of eNOS(-/-) mice as compared with wild-type mice. LMA eNOS(-/-) mice displayed an increased inflammatory phenotype as compared with LMA wild-type mice. Importantly, LMA eNOS(-/-) mice performed worse in a radial arm maze test of spatial learning and memory as compared with LMA wild-type mice. These data suggest that chronic loss of endothelial NO may be an important contributor to both Aβ related pathology and cognitive decline. Cardiovascular risk factors are associated with increased incidence of Alzheimer's disease (AD). A common feature of these risk factors is decreased endothelial nitric oxide (NO). We observed, in mice deficient in endothelial nitric oxide synthase, increased amyloid precursor protein (APP), β-site APP cleaving enzyme 1, amyloid beta levels, microglial activation, and impaired spatial memory. This suggests chronic loss of endothelial NO may be an important contributor to the pathogenesis of sporadic AD.
Keywords: Alzheimer's disease; amyloid precursor protein; endothelium; memory; microglia; nitric oxide.
© 2013 International Society for Neurochemistry.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures




References
-
- Arancio O, Lev-Ram V, Tsien RY, Kandel ER, Hawkins RD. Nitric oxide acts as a retrograde messenger during long-term potentiation in cultured hippocampal neurons. J Physiol Paris. 1996;90:321–322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

