MICU1 controls both the threshold and cooperative activation of the mitochondrial Ca²⁺ uniporter
- PMID: 23747253
- PMCID: PMC3722067
- DOI: 10.1016/j.cmet.2013.04.020
MICU1 controls both the threshold and cooperative activation of the mitochondrial Ca²⁺ uniporter
Abstract
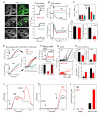
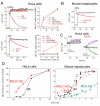
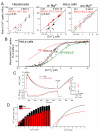
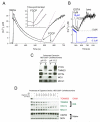
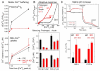
Mitochondrial Ca(2+) uptake via the uniporter is central to cell metabolism, signaling, and survival. Recent studies identified MCU as the uniporter's likely pore and MICU1, an EF-hand protein, as its critical regulator. How this complex decodes dynamic cytoplasmic [Ca(2+)] ([Ca(2+)]c) signals, to tune out small [Ca(2+)]c increases yet permit pulse transmission, remains unknown. We report that loss of MICU1 in mouse liver and cultured cells causes mitochondrial Ca(2+) accumulation during small [Ca(2+)]c elevations but an attenuated response to agonist-induced [Ca(2+)]c pulses. The latter reflects loss of positive cooperativity, likely via the EF-hands. MICU1 faces the intermembrane space and responds to [Ca(2+)]c changes. Prolonged MICU1 loss leads to an adaptive increase in matrix Ca(2+) binding, yet cells show impaired oxidative metabolism and sensitization to Ca(2+) overload. Collectively, the data indicate that MICU1 senses the [Ca(2+)]c to establish the uniporter's threshold and gain, thereby allowing mitochondria to properly decode different inputs.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures
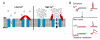
References
-
- Belousov VV, Fradkov AF, Lukyanov KA, Staroverov DB, Shakhbazov KS, Terskikh AV, Lukyanov S. Genetically encoded fluorescent indicator for intracellular hydrogen peroxide. Nature methods. 2006;3:281–286. - PubMed
-
- Beutner G, Sharma VK, Lin L, Ryu SY, Dirksen RT, Sheu SS. Type 1 ryanodine receptor in cardiac mitochondria: Transducer of excitation-metabolism coupling. Biochimica et biophysica acta. 2005 - PubMed
-
- Bragadin M, Pozzan T, Azzone GF. Kinetics of Ca2+ carrier in rat liver mitochondria. Biochemistry. 1979;18:5972–5978. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

