Functional-group-tolerant, nickel-catalyzed cross-coupling reaction for enantioselective construction of tertiary methyl-bearing stereocenters
- PMID: 23751004
- PMCID: PMC3738227
- DOI: 10.1021/ja4034999
Functional-group-tolerant, nickel-catalyzed cross-coupling reaction for enantioselective construction of tertiary methyl-bearing stereocenters
Abstract
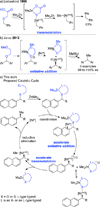
The first Negishi nickel-catalyzed stereospecific cross-coupling reaction of secondary benzylic esters is reported. A series of traceless directing groups is evaluated for ability to promote cross-coupling with dimethylzinc. Esters with a chelating thioether derived from commercially available 2-(methylthio)acetic acid are most effective. The products are formed in high yield and with excellent stereospecificity. A variety of functional groups are tolerated in the reaction including alkenes, alkynes, esters, amines, imides, and O-, S-, and N-heterocycles. The utility of this transformation is highlighted in the enantioselective synthesis of a retinoic acid receptor agonist and a fatty acid amide hydrolase inhibitor.
Figures






References
-
- Barreiro EJ, Kümmerle AE, Fraga CAM. Chem. Rev. 2011;111:5215. - PubMed
-
- Zimmerman J, Buchdunger E, Mett H, Meyer T, Lydon NB, Traxler P. Bioorg. Med. Chem. Lett. 1996;6:1221.
- Zimmerman J, Furet P, Buchdunger E. ACS Symp. Ser. 2001;796:245.
-
-
For examples of methyl substitution to improve metabolic stability of heterocycles, see the following references: Obach RS, Kalgutkar AS, Ryder TF, Walker GS. Chem. Res. Toxicol. 2008;21:1890. Talley JJ, Bertenshaw SR, Brown DL, Carter JS, Graneto MJ, Kellogg MS, Koboldt CM, Yuan J, Zhang YY, Seibert K. J. Med. Chem. 2000;43:1661. Talley JJ, Brown DL, Carter JS, Graneto MJ, Koboldt CM, Masferrer JL, Perkins WE, Rogers RS, Shaffer AF, Zhang YY, Zweifel BS, Seibert K. J. Med. Chem. 2000;43:775.
-
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

