Indoleamine 2,3-dioxygenase is a critical resistance mechanism in antitumor T cell immunotherapy targeting CTLA-4
- PMID: 23752227
- PMCID: PMC3698523
- DOI: 10.1084/jem.20130066
Indoleamine 2,3-dioxygenase is a critical resistance mechanism in antitumor T cell immunotherapy targeting CTLA-4
Abstract
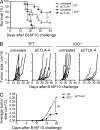
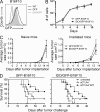
The cytotoxic T lymphocyte antigen-4 (CTLA-4)-blocking antibody ipilimumab results in durable responses in metastatic melanoma, though therapeutic benefit has been limited to a fraction of patients. This calls for identification of resistance mechanisms and development of combinatorial strategies. Here, we examine the inhibitory role of indoleamine 2,3-dioxygenase (IDO) on the antitumor efficacy of CTLA-4 blockade. In IDO knockout mice treated with anti-CTLA-4 antibody, we demonstrate a striking delay in B16 melanoma tumor growth and increased overall survival when compared with wild-type mice. This was also observed with antibodies targeting PD-1-PD-L1 and GITR. To highlight the therapeutic relevance of these findings, we show that CTLA-4 blockade strongly synergizes with IDO inhibitors to mediate rejection of both IDO-expressing and nonexpressing poorly immunogenic tumors, emphasizing the importance of the inhibitory role of both tumor- and host-derived IDO. This effect was T cell dependent, leading to enhanced infiltration of tumor-specific effector T cells and a marked increase in the effector-to-regulatory T cell ratios in the tumors. Overall, these data demonstrate the immunosuppressive role of IDO in the context of immunotherapies targeting immune checkpoints and provide a strong incentive to clinically explore combination therapies using IDO inhibitors irrespective of IDO expression by the tumor cells.
Figures






References
-
- Attia P., Phan G.Q., Maker A.V., Robinson M.R., Quezado M.M., Yang J.C., Sherry R.M., Topalian S.L., Kammula U.S., Royal R.E., et al. 2005. Autoimmunity correlates with tumor regression in patients with metastatic melanoma treated with anti-cytotoxic T-lymphocyte antigen-4. J. Clin. Oncol. 23:6043–6053 10.1200/JCO.2005.06.205 - DOI - PMC - PubMed
-
- Balachandran V.P., Cavnar M.J., Zeng S., Bamboat Z.M., Ocuin L.M., Obaid H., Sorenson E.C., Popow R., Ariyan C., Rossi F., et al. 2011. Imatinib potentiates antitumor T cell responses in gastrointestinal stromal tumor through the inhibition of Ido. Nat. Med. 17:1094–1100 10.1038/nm.2438 - DOI - PMC - PubMed
-
- Belladonna M.L., Grohmann U., Guidetti P., Volpi C., Bianchi R., Fioretti M.C., Schwarcz R., Fallarino F., Puccetti P. 2006. Kynurenine pathway enzymes in dendritic cells initiate tolerogenesis in the absence of functional IDO. J. Immunol. 177:130–137 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

