Regulation of cell division and expansion by sugar and auxin signaling
- PMID: 23755057
- PMCID: PMC3667240
- DOI: 10.3389/fpls.2013.00163
Regulation of cell division and expansion by sugar and auxin signaling
Abstract
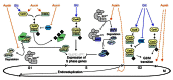
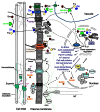
Plant growth and development are modulated by concerted actions of a variety of signaling molecules. In recent years, evidence has emerged on the roles of sugar and auxin signals network in diverse aspects of plant growth and development. Here, based on recent progress of genetic analyses and gene expression profiling studies, we summarize the functional similarities, diversities, and their interactions of sugar and auxin signals in regulating two major processes of plant development: cell division and cell expansion. We focus on roles of sugar and auxin signaling in both vegetative and reproductive tissues including developing seed.
Keywords: auxin signaling; cell division; cell expansion; seed development; sugar signaling.
Figures
Similar articles
-
Role of sugar and auxin crosstalk in plant growth and development.Physiol Plant. 2022 Jan;174(1):e13546. doi: 10.1111/ppl.13546. Epub 2021 Sep 17. Physiol Plant. 2022. PMID: 34480799 Review.
-
Cellular responses to auxin: division versus expansion.Cold Spring Harb Perspect Biol. 2010 May;2(5):a001446. doi: 10.1101/cshperspect.a001446. Epub 2010 Apr 7. Cold Spring Harb Perspect Biol. 2010. PMID: 20452959 Free PMC article. Review.
-
The MEDIATOR genes MED12 and MED13 control Arabidopsis root system configuration influencing sugar and auxin responses.Plant Mol Biol. 2017 Sep;95(1-2):141-156. doi: 10.1007/s11103-017-0647-z. Epub 2017 Aug 5. Plant Mol Biol. 2017. PMID: 28780645
-
A genomics approach to understanding the role of auxin in apple (Malus x domestica) fruit size control.BMC Plant Biol. 2012 Jan 13;12:7. doi: 10.1186/1471-2229-12-7. BMC Plant Biol. 2012. PMID: 22243694 Free PMC article.
-
Mechanisms regulating auxin action during fruit development.Physiol Plant. 2014 May;151(1):62-72. doi: 10.1111/ppl.12142. Epub 2014 Jan 29. Physiol Plant. 2014. PMID: 24329770
Cited by
-
Effects of Polyploidization on Morphology, Photosynthetic Parameters and Sucrose Metabolism in Lily.Plants (Basel). 2022 Aug 14;11(16):2112. doi: 10.3390/plants11162112. Plants (Basel). 2022. PMID: 36015415 Free PMC article.
-
The Role of Auxin in Cell Wall Expansion.Int J Mol Sci. 2018 Mar 22;19(4):951. doi: 10.3390/ijms19040951. Int J Mol Sci. 2018. PMID: 29565829 Free PMC article. Review.
-
Cucurbitaceae genome evolution, gene function and molecular breeding.Hortic Res. 2022 Jan 19;9:uhab057. doi: 10.1093/hr/uhab057. Online ahead of print. Hortic Res. 2022. PMID: 35043161 Free PMC article.
-
Comparative Metabolomics Profiling Reveals Key Metabolites and Associated Pathways Regulating Tuber Dormancy in White Yam (Dioscorea rotundata Poir.).Metabolites. 2023 Apr 28;13(5):610. doi: 10.3390/metabo13050610. Metabolites. 2023. PMID: 37233651 Free PMC article.
-
Transcriptomic analysis of short-fruit 1 (sf1) reveals new insights into the variation of fruit-related traits in Cucumis sativus.Sci Rep. 2017 Jun 7;7(1):2950. doi: 10.1038/s41598-017-02932-5. Sci Rep. 2017. PMID: 28592854 Free PMC article.
References
-
- Atif R. M., Boulisset F., Conreux C., Thompson R., Ochatt S. J. (2012). In vitro auxin treatment promotes cell division and delays endoreduplication in developing seeds of the model legume species Medicago truncatula. Physiol. Plant. 10.1111/j.1399-3054.2012.01719.x[Epubaheadofprint]. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

