Phase-transition thresholds and vaporization phenomena for ultrasound phase-change nanoemulsions assessed via high-speed optical microscopy
- PMID: 23760161
- PMCID: PMC4703326
- DOI: 10.1088/0031-9155/58/13/4513
Phase-transition thresholds and vaporization phenomena for ultrasound phase-change nanoemulsions assessed via high-speed optical microscopy
Abstract
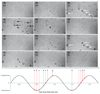
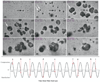
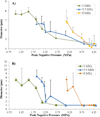
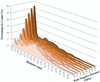
Ultrasonically activated phase-change contrast agents (PCCAs) based on perfluorocarbon droplets have been proposed for a variety of therapeutic and diagnostic clinical applications. When generated at the nanoscale, droplets may be small enough to exit the vascular space and then be induced to vaporize with high spatial and temporal specificity by externally-applied ultrasound. The use of acoustical techniques for optimizing ultrasound parameters for given applications can be a significant challenge for nanoscale PCCAs due to the contributions of larger outlier droplets. Similarly, optical techniques can be a challenge due to the sub-micron size of nanodroplet agents and resolution limits of optical microscopy. In this study, an optical method for determining activation thresholds of nanoscale emulsions based on the in vitro distribution of bubbles resulting from vaporization of PCCAs after single, short (<10 cycles) ultrasound pulses is evaluated. Through ultra-high-speed microscopy it is shown that the bubbles produced early in the pulse from vaporized droplets are strongly affected by subsequent cycles of the vaporization pulse, and these effects increase with pulse length. Results show that decafluorobutane nanoemulsions with peak diameters on the order of 200 nm can be optimally vaporized with short pulses using pressures amenable to clinical diagnostic ultrasound machines.
Figures






Similar articles
-
Image-Guided Ultrasound Characterization of Volatile Sub-Micron Phase-Shift Droplets in the 20-40 MHz Frequency Range.Ultrasound Med Biol. 2016 Mar;42(3):795-807. doi: 10.1016/j.ultrasmedbio.2015.11.012. Epub 2015 Dec 24. Ultrasound Med Biol. 2016. PMID: 26725168
-
Quantifying activation of perfluorocarbon-based phase-change contrast agents using simultaneous acoustic and optical observation.Ultrasound Med Biol. 2015 May;41(5):1422-31. doi: 10.1016/j.ultrasmedbio.2014.12.021. Epub 2015 Feb 2. Ultrasound Med Biol. 2015. PMID: 25656747 Free PMC article.
-
Design of ultrasonically-activatable nanoparticles using low boiling point perfluorocarbons.Biomaterials. 2012 Apr;33(11):3262-9. doi: 10.1016/j.biomaterials.2012.01.021. Epub 2012 Jan 29. Biomaterials. 2012. PMID: 22289265 Free PMC article.
-
Phase-change contrast agents for imaging and therapy.Curr Pharm Des. 2012;18(15):2152-65. doi: 10.2174/138161212800099883. Curr Pharm Des. 2012. PMID: 22352770 Free PMC article. Review.
-
Optimizing Acoustic Activation of Phase Change Contrast Agents With the Activation Pressure Matching Method: A Review.IEEE Trans Ultrason Ferroelectr Freq Control. 2017 Jan;64(1):264-272. doi: 10.1109/TUFFC.2016.2616304. Epub 2016 Oct 12. IEEE Trans Ultrason Ferroelectr Freq Control. 2017. PMID: 27740481 Free PMC article. Review.
Cited by
-
Vaporization dynamics of volatile perfluorocarbon droplets: a theoretical model and in vitro validation.Med Phys. 2014 Oct;41(10):102901. doi: 10.1118/1.4894804. Med Phys. 2014. PMID: 25281982 Free PMC article.
-
Phase-Conversion Nanoparticle Contrast Agents: Do Good Things Come in Small Packages?Circ Cardiovasc Imaging. 2016 Jan;9(1):10.1161/CIRCIMAGING.115.004374 e004374. doi: 10.1161/CIRCIMAGING.115.004374. Circ Cardiovasc Imaging. 2016. PMID: 26712163 Free PMC article. No abstract available.
-
Initial nucleation of nanodroplets in viscoelastic tissue driven by ultrasound: A theoretical simulation.Ultrason Sonochem. 2025 Apr;115:107285. doi: 10.1016/j.ultsonch.2025.107285. Epub 2025 Feb 24. Ultrason Sonochem. 2025. PMID: 40037141 Free PMC article.
-
Size-Sorted Superheated Nanodroplets for Dosimetry and Range Verification of Carbon-Ion Radiotherapy.Nanomaterials (Basel). 2024 Oct 13;14(20):1643. doi: 10.3390/nano14201643. Nanomaterials (Basel). 2024. PMID: 39452979 Free PMC article.
-
Standing wave-assisted acoustic droplet vaporization for single and dual payload release in acoustically-responsive scaffolds.Ultrason Sonochem. 2020 Sep;66:105109. doi: 10.1016/j.ultsonch.2020.105109. Epub 2020 Mar 26. Ultrason Sonochem. 2020. PMID: 32248042 Free PMC article.
References
-
- Brennen CE. Cavitation and Bubble Dynamics. Oxford, New York: Oxford University Press; 1995.
-
- Campbell RB. Tumor physiology and delivery of nanopharmaceuticals. Anticancer Agents Med Chem. 2006;6:503–512. - PubMed
-
- Cobbold RSC. Foundations of biomedical ultrasound. New York, New York: Oxford University Press; 2007.
-
- Couture O, Faivre M, Pannacci N, Babataheri A, Servois V, Tabeling P, Tanter M. Ultrasound internal tattooing. Medical Physics. 2011;38:1116–1123. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
