Yes-associated protein (YAP) increases chemosensitivity of hepatocellular carcinoma cells by modulation of p53
- PMID: 23760493
- PMCID: PMC3813567
- DOI: 10.4161/cbt.24345
Yes-associated protein (YAP) increases chemosensitivity of hepatocellular carcinoma cells by modulation of p53
Abstract
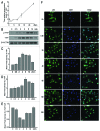
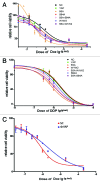
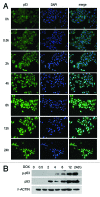
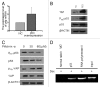
The yes-associated protein (YAP) transcription co-activator has been reported either as an oncogene candidate or a tumor suppressor. Liver tissue chips revealed that about 51.4% human hepatocellular carcinoma (HCC) samples express YAP and 32.9% HCC samples express phosphorylated YAP. In this study, we found that chemotherapy increased YAP protein expression and nuclear translocation in HepG2 cells, as well as p53 protein expression and nuclear translocation. However, little is known about YAP functions during chemotherapy. Our results show that overexpression of YAP increases chemosensitivity of HepG2 cells during chemotherapy. Dominant negative transfection of Flag-S94A (TEAD binding domain mutant) or Flag-W1W2 (WW domain mutant) to HepG2 cells decreases p53 expression/ nuclear translocation and chemosensitivity when compared with control HepG2 cells. Furthermore, rescue transfection of Flag-5SA-S94A or Flag-5SA-W1W2, respectively to HepG2 cells regains p53 expression/nuclear translocation and chemosensitivity. These results indicate that YAP promotes chemosensitivity by modulating p53 during chemotherapy and both TEAD and WW binding domains are required for YAP-mediated p53 function. ChIP assay results also indicated that YAP binds directly to the p53 promoter to improve its expression. In addition, p53 could positively feedback YAP expression through binding to the YAP promoter. Taken together, our current data indicate that YAP functions as a tumor suppressor that enhances apoptosis by modulating p53 during chemotherapy.
Keywords: YAP; hepatocellular carcinoma; p53.
Figures



References
-
- Trinchet JC, Beaugrand M. [Hepatocellular carcinoma: treatment with arterial chemo-embolization] Presse Med. 1994;23:831–3. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
