Amyloid precursor proteins interact with the heterotrimeric G protein Go in the control of neuronal migration
- PMID: 23761911
- PMCID: PMC3682380
- DOI: 10.1523/JNEUROSCI.1146-13.2013
Amyloid precursor proteins interact with the heterotrimeric G protein Go in the control of neuronal migration
Abstract
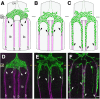
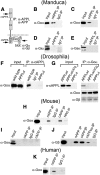
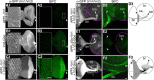
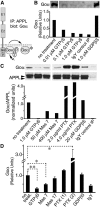
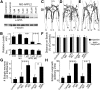
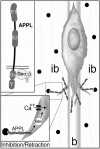
Amyloid precursor protein (APP) belongs to a family of evolutionarily conserved transmembrane glycoproteins that has been proposed to regulate multiple aspects of cell motility in the nervous system. Although APP is best known as the source of β-amyloid fragments (Aβ) that accumulate in Alzheimer's disease, perturbations affecting normal APP signaling events may also contribute to disease progression. Previous in vitro studies showed that interactions between APP and the heterotrimeric G protein Goα-regulated Goα activity and Go-dependent apoptotic responses, independent of Aβ. However, evidence for authentic APP-Go interactions within the healthy nervous system has been lacking. To address this issue, we have used a combination of in vitro and in vivo strategies to show that endogenously expressed APP family proteins colocalize with Goα in both insect and mammalian nervous systems, including human brain. Using biochemical, pharmacological, and Bimolecular Fluorescence Complementation assays, we have shown that insect APP (APPL) directly interacts with Goα in cell culture and at synaptic terminals within the insect brain, and that this interaction is regulated by Goα activity. We have also adapted a well characterized assay of neuronal migration in the hawkmoth Manduca to show that perturbations affecting APPL and Goα signaling induce the same unique pattern of ectopic, inappropriate growth and migration, analogous to defective migration patterns seen in mice lacking all APP family proteins. These results support the model that APP and its orthologs regulate conserved aspects of neuronal migration and outgrowth in the nervous system by functioning as unconventional Goα-coupled receptors.
Figures



References
-
- Banker G, Goslin K. Culturing nerve cells. Cambridge, MA: MIT; 1998.
-
- Bergmans BA, Shariati SA, Habets RL, Verstreken P, Schoonjans L, Müller U, Dotti CG, De Strooper B. Neurons generated from APP/APLP1/APLP2 triple knockout embryonic stem cells behave normally in vitro and in vivo: lack of evidence for a cell autonomous role of the amyloid precursor protein in neuronal differentiation. Stem Cells. 2010;28:399–406. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
