Mate choice for genetic compatibility in the house mouse
- PMID: 23762510
- PMCID: PMC3678478
- DOI: 10.1002/ece3.534
Mate choice for genetic compatibility in the house mouse
Abstract
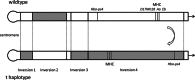
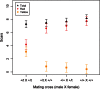
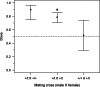
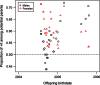
In house mice, genetic compatibility is influenced by the t haplotype, a driving selfish genetic element with a recessive lethal allele, imposing fundamental costs on mate choice decisions. Here, we evaluate the cost of genetic incompatibility and its implication for mate choice in a wild house mice population. In laboratory reared mice, we detected no fertility (number of embryos) or fecundity (ability to conceive) costs of the t, and yet we found a high cost of genetic incompatibility: heterozygote crosses produced 40% smaller birth litter sizes because of prenatal mortality. Surprisingly, transmission of t in crosses using +/t males was influenced by female genotype, consistent with postcopulatory female choice for + sperm in +/t females. Analysis of paternity patterns in a wild population of house mice showed that +/t females were more likely than +/+ females to have offspring sired by +/+ males, and unlike +/+ females, paternity of their offspring was not influenced by +/t male frequency, further supporting mate choice for genetic compatibility. As the major histocompatibility complex (MHC) is physically linked to the t, we investigated whether females could potentially use variation at the MHC to identify male genotype at the sperm or individual level. A unique MHC haplotype is linked to the t haplotype. This MHC haplotype could allow the recognition of t and enable pre- and postcopulatory mate choice for genetic compatibility. Alternatively, the MHC itself could be the target of mate choice for genetic compatibility. We predict that mate choice for genetic compatibility will be difficult to find in many systems, as only weak fertilization biases were found despite an exceptionally high cost of genetic incompatibility.
Keywords: Cryptic female choice; meiotic drive; segregation distortion; selection arena hypothesis; sexual selection; sperm selection; t-complex.
Figures


Similar articles
-
No evidence for female discrimination against male house mice carrying a selfish genetic element.Curr Zool. 2016 Dec;62(6):675-685. doi: 10.1093/cz/zow063. Epub 2016 Jun 25. Curr Zool. 2016. PMID: 29491955 Free PMC article.
-
Female house mice avoid fertilization by t haplotype incompatible males in a mate choice experiment.J Evol Biol. 2015 Jan;28(1):54-64. doi: 10.1111/jeb.12525. Epub 2014 Dec 14. J Evol Biol. 2015. PMID: 25494878 Free PMC article.
-
Meiotic drive changes sperm precedence patterns in house mice: potential for male alternative mating tactics?BMC Evol Biol. 2016 Jun 21;16(1):133. doi: 10.1186/s12862-016-0710-4. BMC Evol Biol. 2016. PMID: 27328665 Free PMC article.
-
Why do females mate multiply? A review of the genetic benefits.Biol Rev Camb Philos Soc. 2000 Feb;75(1):21-64. doi: 10.1017/s0006323199005423. Biol Rev Camb Philos Soc. 2000. PMID: 10740892 Review.
-
Genetic compatibility, mate choice and patterns of parentage: invited review.Mol Ecol. 2000 Aug;9(8):1013-27. doi: 10.1046/j.1365-294x.2000.00964.x. Mol Ecol. 2000. PMID: 10964221 Review.
Cited by
-
Unexpected patterns of segregation distortion at a selfish supergene in the fire ant Solenopsis invicta.BMC Genet. 2018 Nov 7;19(1):101. doi: 10.1186/s12863-018-0685-9. BMC Genet. 2018. PMID: 30404617 Free PMC article.
-
Meiotic drive adaptive testes enlargement during early development in the stalk-eyed fly.Biol Lett. 2022 Nov;18(11):20220352. doi: 10.1098/rsbl.2022.0352. Epub 2022 Nov 30. Biol Lett. 2022. PMID: 36448294 Free PMC article.
-
Meiotic drive in house mice: mechanisms, consequences, and insights for human biology.Chromosome Res. 2022 Sep;30(2-3):165-186. doi: 10.1007/s10577-022-09697-2. Epub 2022 Jul 13. Chromosome Res. 2022. PMID: 35829972 Free PMC article. Review.
-
The clock gene Period1 regulates innate routine behaviour in mice.Proc Biol Sci. 2014 Mar 5;281(1781):20140034. doi: 10.1098/rspb.2014.0034. Print 2014 Apr 22. Proc Biol Sci. 2014. PMID: 24598427 Free PMC article.
-
No evidence for female discrimination against male house mice carrying a selfish genetic element.Curr Zool. 2016 Dec;62(6):675-685. doi: 10.1093/cz/zow063. Epub 2016 Jun 25. Curr Zool. 2016. PMID: 29491955 Free PMC article.
References
-
- Amadou C, Younger RM, Sims S, Matthews LH, Rogers J, Kumanovics A, et al. Co-duplication of olfactory receptor and MHC class I genes in the mouse major histocompatibility complex. Hum. Mol. Genet. 2003;12:3025–3040. - PubMed
-
- Andersson M, Simmons LW. Sexual selection and mate choice. Trends Ecol. Evol. 2006;21:296–302. - PubMed
-
- Apanius V, Penn D, Slev PR, Ruff LR, Potts WK. The nature of selection on the major histocompatibility complex. Crit. Rev. Immunol. 1997;17:179–224. [Review] - PubMed
-
- Ardlie K, Silver LM. Recent evolution of mouse t haplotypes at polymorphic microsatellites associated with the t complex responder (Tcr) locus. Genet. Res. 1996;67:1–10. - PubMed
-
- Artzt K. Gene mapping within the T/t complex of the mouse III: t-lethal genes are arranged in three clusters on chromosome 17. Cell. 1984;39:565–572. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

