The thalamostriatal pathway and cholinergic control of goal-directed action: interlacing new with existing learning in the striatum
- PMID: 23770257
- PMCID: PMC3863609
- DOI: 10.1016/j.neuron.2013.04.039
The thalamostriatal pathway and cholinergic control of goal-directed action: interlacing new with existing learning in the striatum
Abstract
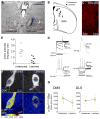
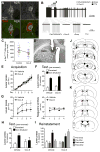
The capacity for goal-directed action depends on encoding specific action-outcome associations, a learning process mediated by the posterior dorsomedial striatum (pDMS). In a changing environment, plasticity has to remain flexible, requiring interference between new and existing learning to be minimized, yet it is not known how new and existing learning are interlaced in this way. Here we investigated the role of the thalamostriatal pathway linking the parafascicular thalamus (Pf) with cholinergic interneurons (CINs) in the pDMS in this process. Removing the excitatory input from Pf to the CINs was found to reduce the firing rate and intrinsic activity of these neurons and produced an enduring deficit in goal-directed learning after changes in the action-outcome contingency. Disconnection of the Pf-pDMS pathway produced similar behavioral effects. These data suggest that CINs reduce interference between new and existing learning, consistent with claims that the thalamostriatal pathway exerts state control over learning-related plasticity.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures




Comment in
-
How did the chicken cross the road? With her striatal cholinergic interneurons, of course.Neuron. 2013 Jul 10;79(1):3-6. doi: 10.1016/j.neuron.2013.06.033. Neuron. 2013. PMID: 23849192 Free PMC article.
References
-
- Apicella P. Leading tonically active neurons of the striatum from reward detection to context recognition. Trends Neurosci. 2007;30:299–306. - PubMed
-
- Balleine BW, Dickinson A. Goal-directed instrumental action: contingency and incentive learning and their cortical substrates. Neuropharmacology. 1998;37:407–419. - PubMed
-
- Balleine BW, Liljeholm M, Ostlund SB. The integrative function of the basal ganglia in instrumental conditioning. Behav Brain Res. 2009;199:43–52. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

