Smoke carcinogens cause bone loss through the aryl hydrocarbon receptor and induction of Cyp1 enzymes
- PMID: 23776235
- PMCID: PMC3704019
- DOI: 10.1073/pnas.1220919110
Smoke carcinogens cause bone loss through the aryl hydrocarbon receptor and induction of Cyp1 enzymes
Abstract
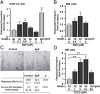
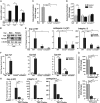
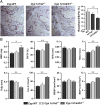
Smoking is a major risk factor for osteoporosis and fracture, but the mechanism through which smoke causes bone loss remains unclear. Here, we show that the smoke toxins benzo(a)pyrene (BaP) and 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) interact with the aryl hydrocarbon receptor (Ahr) to induce osteoclastic bone resorption through the activation of cytochrome P450 1a/1b (Cyp1) enzymes. BaP and TCDD enhanced osteoclast formation in bone marrow cell cultures and gavage with BaP stimulated bone resorption and osteoclastogenesis in vivo. The osteoclastogenesis triggered by BaP or RANK-L was reduced in Ahr(-/-) cells, consistent with the high bone mass noted in Ahr(-/-) male mice. The receptor activator of NF-κB ligand (RANK-L) also failed to induce the expression of Cyp1 enzymes in Ahr(-/-) cells. Furthermore, the osteoclastogenesis induced by TCDD was lower in Cyp1a1/1a2(-/-) and Cyp1a1/1a2/1b1(-/-) cultures, indicating that Ahr was upstream of the Cyp enzymes. Likewise, the pharmacological inhibition of the Cyp1 enzymes with tetramethylsilane or proadifen reduced osteoclastogenesis. Finally, deletion of the Cyp1a1, Cyp1a2, and Cyp1b1 in triple knockout mice resulted in reduced bone resorption and recapitulated the high bone mass phenotype of Ahr(-/-) mice. Overall, the data identify the Ahr and Cyp1 enzymes not only in the pathophysiology of smoke-induced osteoporosis, but also as potential targets for selective modulation by new therapeutics.
Keywords: bone formation; osteoblast; skeletal remodeling; toxicology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Wipfli H, Samet JM. Global economic and health benefits of tobacco control: Part 1. Clin Pharmacol Ther. 2009;86(3):263–271. - PubMed
-
- World Health Organization Collaborating Centre for Metabolic Bone Disease. (2013) FRAX (University of Sheffield, Sheffield, UK). Available at www.shef.ac.uk/FRAX.
-
- Kanis JA, et al. Smoking and fracture risk: A meta-analysis. Osteoporos Int. 2005;16(2):155–162. - PubMed
-
- Szulc P, et al. Increased bone resorption in moderate smokers with low body weight: The Minos study. J Clin Endocrinol Metab. 2002;87(2):666–674. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- AG40132/AG/NIA NIH HHS/United States
- AG23176/AG/NIA NIH HHS/United States
- R01 AG040132/AG/NIA NIH HHS/United States
- ES08147/ES/NIEHS NIH HHS/United States
- DK80459/DK/NIDDK NIH HHS/United States
- R01 ES014403/ES/NIEHS NIH HHS/United States
- P30 ES006096/ES/NIEHS NIH HHS/United States
- CA22763/CA/NCI NIH HHS/United States
- P30 CA016520/CA/NCI NIH HHS/United States
- P30 ES06096/ES/NIEHS NIH HHS/United States
- R01 GM034883/GM/NIGMS NIH HHS/United States
- GM34883/GM/NIGMS NIH HHS/United States
- ES014403/ES/NIEHS NIH HHS/United States
- R01 DK080459/DK/NIDDK NIH HHS/United States
- R01 AG023176/AG/NIA NIH HHS/United States
- R01 ES008147/ES/NIEHS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

