Sympathetic neurons and chromaffin cells share a common progenitor in the neural crest in vivo
- PMID: 23777568
- PMCID: PMC3693940
- DOI: 10.1186/1749-8104-8-12
Sympathetic neurons and chromaffin cells share a common progenitor in the neural crest in vivo
Abstract
Background: The neural crest (NC) is a transient embryonic structure unique to vertebrates, which generates peripheral sensory and autonomic neurons, glia, neuroendocrine chromaffin and thyroid C-cells, melanocytes, and mesenchymal derivatives such as parts of the skull, heart, and meninges. The sympathoadrenal (SA) cell lineage is one major sub-lineage of the NC that gives rise to sympathetic neurons, chromaffin cells, and the intermediate small intensely fluorescent (SIF) cells. A key question is when during NC ontogeny do multipotent progenitors segregate into the different NC-derived lineages. Recent evidence suggested that sympathetic, sensory, and melanocyte progenitors delaminate from the thoracic neural tube (NT) in successive, largely non-overlapping waves and that at least certain NC progenitors are already fate-restricted within the NT. Whether sympathetic neurons and chromaffin cells, suggested by cell culture studies to share a common progenitor, are also fate segregated in ovo prior to emigration, is not known.
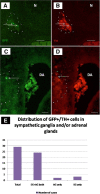
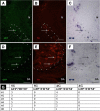
Results: We have conducted single cell electroporations of a GFP-encoding plasmid into the dorsal midline of E2 chick NTs at the adrenomedullary level of the NC. Analysis of their derivatives, performed at E6, revealed that in most cases, labelled progeny was detected in both sympathetic ganglia and adrenal glands, where cells co-expressed characteristic marker combinations.
Conclusions: Our results show that sympathetic neurons and adrenal chromaffin cells share a common progenitor in the NT. Together with previous findings we suggest that phenotypic diversification of these sublineages is likely to occur after delamination from the NT and prior to target encounter.
Figures

Similar articles
-
The sympathoadrenal cell lineage: specification, diversification, and new perspectives.Dev Biol. 2006 Oct 15;298(2):335-43. doi: 10.1016/j.ydbio.2006.07.010. Epub 2006 Jul 14. Dev Biol. 2006. PMID: 16928368 Review.
-
The development of the chromaffin cell lineage from the neural crest.Auton Neurosci. 2009 Nov 17;151(1):10-6. doi: 10.1016/j.autneu.2009.07.020. Epub 2009 Aug 14. Auton Neurosci. 2009. PMID: 19683477 Review.
-
Generation of neuroendocrine chromaffin cells from sympathoadrenal progenitors: beyond the glucocorticoid hypothesis.Ann N Y Acad Sci. 2002 Oct;971:554-9. doi: 10.1111/j.1749-6632.2002.tb04526.x. Ann N Y Acad Sci. 2002. PMID: 12438182 Review.
-
Molecular control of cell fate in the neural crest: the sympathoadrenal lineage.Annu Rev Neurosci. 1993;16:129-58. doi: 10.1146/annurev.ne.16.030193.001021. Annu Rev Neurosci. 1993. PMID: 8460888 Review.
-
Cell fate decisions during neural crest ontogeny.Int J Dev Biol. 2017;61(3-4-5):195-203. doi: 10.1387/ijdb.160196ck. Int J Dev Biol. 2017. PMID: 28621417 Review.
Cited by
-
Induced pluripotent stem cells for disease modeling, cell therapy and drug discovery in genetic autonomic disorders: a review.Clin Auton Res. 2019 Aug;29(4):367-384. doi: 10.1007/s10286-018-00587-4. Epub 2019 Jan 10. Clin Auton Res. 2019. PMID: 30631982 Review.
-
Generation of Adrenal Chromaffin-like Cells from Human Pluripotent Stem Cells.Stem Cell Reports. 2018 Jan 9;10(1):134-150. doi: 10.1016/j.stemcr.2017.11.003. Epub 2017 Dec 7. Stem Cell Reports. 2018. PMID: 29233551 Free PMC article.
-
Cancer Stem Cells in Pheochromocytoma and Paraganglioma.Front Endocrinol (Lausanne). 2020 Feb 25;11:79. doi: 10.3389/fendo.2020.00079. eCollection 2020. Front Endocrinol (Lausanne). 2020. PMID: 32158431 Free PMC article. Review.
-
Defining Pathological Activities of ALK in Neuroblastoma, a Neural Crest-Derived Cancer.Int J Mol Sci. 2021 Oct 29;22(21):11718. doi: 10.3390/ijms222111718. Int J Mol Sci. 2021. PMID: 34769149 Free PMC article. Review.
-
Stem Cells, Self-Renewal, and Lineage Commitment in the Endocrine System.Front Endocrinol (Lausanne). 2019 Nov 8;10:772. doi: 10.3389/fendo.2019.00772. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 31781041 Free PMC article. Review.
References
-
- LeDouarin NM, Kalcheim C. The Neural Crest. Cambridge: Cambridge University Press; 1999.
-
- Unsicker K, Huber K, Schütz G, Kalcheim C. The chromaffin cell and its development. Neurochem Res. 2005;30(6–7):921–925. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

