Interactions between Asaia, Plasmodium and Anopheles: new insights into mosquito symbiosis and implications in malaria symbiotic control
- PMID: 23777746
- PMCID: PMC3708832
- DOI: 10.1186/1756-3305-6-182
Interactions between Asaia, Plasmodium and Anopheles: new insights into mosquito symbiosis and implications in malaria symbiotic control
Abstract
Background: Malaria represents one of the most devastating infectious diseases. The lack of an effective vaccine and the emergence of drug resistance make necessary the development of new effective control methods. The recent identification of bacteria of the genus Asaia, associated with larvae and adults of malaria vectors, designates them as suitable candidates for malaria paratransgenic control.To better characterize the interactions between Asaia, Plasmodium and the mosquito immune system we performed an integrated experimental approach.
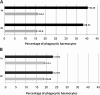
Methods: Quantitative PCR analysis of the amount of native Asaia was performed on individual Anopheles stephensi specimens. Mosquito infection was carried out with the strain PbGFPCON and the number of parasites in the midgut was counted by fluorescent microscopy.The colonisation of infected mosquitoes was achieved using GFP or DsRed tagged-Asaia strains.Reverse transcriptase-PCR analysis, growth and phagocytosis tests were performed using An. stephensi and Drosophila melanogaster haemocyte cultures and DsRed tagged-Asaia and Escherichia coli strains.
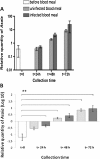
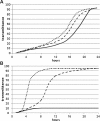
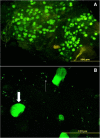
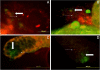
Results: Using quantitative PCR we have quantified the relative amount of Asaia in infected and uninfected mosquitoes, showing that the parasite does not interfere with bacterial blooming. The correlation curves have confirmed the active replication of Asaia, while at the same time, the intense decrease of the parasite.The 'in vitro' immunological studies have shown that Asaia induces the expression of antimicrobial peptides, however, the growth curves in conditioned medium as well as a phagocytosis test, indicated that the bacterium is not an immune-target.Using fluorescent strains of Asaia and Plasmodium we defined their co-localisation in the mosquito midgut and salivary glands.
Conclusions: We have provided important information about the relationship of Asaia with both Plasmodium and Anopheles. First, physiological changes in the midgut following an infected or uninfected blood meal do not negatively affect the residing Asaia population that seems to benefit from this condition. Second, Asaia can act as an immune-modulator activating antimicrobial peptide expression and seems to be adapted to the host immune response. Last, the co-localization of Asaia and Plasmodium highlights the possibility of reducing vectorial competence using bacterial recombinant strains capable of releasing anti-parasite molecules.
Figures


References
-
- Sinden RE. Plasmodium differentiation in the mosquito. Parassitologia. 1999;41:139–148. - PubMed
-
- Sinden RE, Alavi Y, Raine JD. Mosquito-malaria interactions: a reappraisal of the concepts of susceptibility and refractoriness. Insect Biochem Mol Biol. 2004;34:625–629. - PubMed
-
- Vaughan JA. Population dynamics of Plasmodium sporogony. Trends Parasitol. 2007;23:63–70. - PubMed
-
- Taylor LH. Infection rates in, and the number of Plasmodiumfalciparum genotypes carried by Anopheles mosquitoes in Tanzania. Ann Trop Med Parasitol. 1999;93:659–662. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

