A randomized, double-blind study of repeated incobotulinumtoxinA (Xeomin(®)) in cervical dystonia
- PMID: 23779062
- PMCID: PMC3834167
- DOI: 10.1007/s00702-013-1048-3
A randomized, double-blind study of repeated incobotulinumtoxinA (Xeomin(®)) in cervical dystonia
Abstract
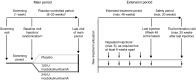
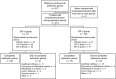
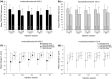
IncobotulinumtoxinA (Xeomin(®), NT 201), a preparation without accessory (complexing) proteins, has shown comparable efficacy and safety to onabotulinumtoxinA in treating cervical dystonia (CD). This study evaluated the efficacy and safety of repeated incobotulinumtoxinA injections in subjects with CD. Following a ≤20-week placebo-controlled, randomized, double-blind, single-dose main period, subjects could enter a ≤68-week prospective, randomized, double-blind, repeated-dose, flexible-interval (minimum 6 weeks) extension period with 240 U or 120 U of incobotulinumtoxinA (≤5 injections). Outcome measures included the Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) and adverse events (AEs). Of 219 subjects completing the main period, 214 were randomized in the extension period to receive incobotulinumtoxinA 240 U (n = 111) or 120 U (n = 103); 169 subjects completed the extension period, with 90 receiving five injection sessions. Both doses of incobotulinumtoxinA provided statistically significant and clinically relevant improvements in mean TWSTRS-Total, -Severity, -Disability, and -Pain scores, from each injection session to respective 4-week follow-up visits. The most frequently reported AE was dysphagia (240 U: 23.4 %; 120 U: 12.6 %), which did not result in any study withdrawals. There was no impact of injection interval on the incidence of AEs (post hoc analysis). A major limitation of this study was the fixed dose design requested by regulatory authorities, which does not reflect clinical practice. However, repeated incobotulinumtoxinA injections (240 or 120 U; flexible intervals) provided sustained efficacy and were well tolerated, with no unexpected safety risks following repeated injections. The incidence of AEs was similar in subjects requiring repeated injections at shorter intervals (≤12 weeks) compared with those treated using longer intervals (>12 weeks).
Figures
References
-
- Allergan Inc. (2011) Botox® US Prescribing Information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/103000s5232lbl.pdf. Accessed 18 June 2012
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

