Cytometric sorting based on the fluorescence lifetime of spectrally overlapping signals
- PMID: 23787669
- PMCID: PMC3726248
- DOI: 10.1364/OE.21.014816
Cytometric sorting based on the fluorescence lifetime of spectrally overlapping signals
Abstract
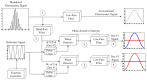
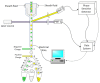
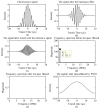
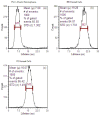
Flow cytometry is a well-established and powerful high- throughput fluorescence measurement tool that also allows for the sorting and enrichment of subpopulations of cells expressing unique fluorescence signatures. Owing to the reliance on intensity-only signals, flow cytometry sorters cannot easily discriminate between fluorophores that spectrally overlap. In this paper we demonstrate a new method of cell sorting using a fluorescence lifetime-dependent methodology. This approach, referred to herein as phase-filtered cell sorting (PFCS), permits sorting based on the average fluorescence lifetime of a fluorophore by separating fluorescence signals from species that emit differing average fluorescence lifetimes. Using lifetime-dependent hardware, cells and microspheres labeled with fluorophores were sorted with purities up to 90%. PFCS is a practical approach for separating populations of cells that are stained with spectrally overlapping fluorophores or that have interfering autofluorescence signals.
Figures




Similar articles
-
Two-color, double-slit in vivo flow cytometer.Opt Lett. 2007 Oct 15;32(20):2993-5. doi: 10.1364/ol.32.002993. Opt Lett. 2007. PMID: 17938677
-
Fluorescence-lifetime imaging with a multifocal two-photon microscope.Opt Lett. 2004 Dec 15;29(24):2884-6. doi: 10.1364/ol.29.002884. Opt Lett. 2004. PMID: 15645812
-
Simultaneous two-photon spectral and lifetime fluorescence microscopy.Appl Opt. 2004 Sep 20;43(27):5173-82. doi: 10.1364/ao.43.005173. Appl Opt. 2004. PMID: 15473237
-
[Nonlinear Microscopy in Ophthalmology: Principles and Pathbreaking Applications].Klin Monbl Augenheilkd. 2015 Dec;232(12):1365-73. doi: 10.1055/s-0041-109020. Epub 2015 Dec 17. Klin Monbl Augenheilkd. 2015. PMID: 26678898 Review. German.
-
Multiphoton fluorescence lifetime contrast in deep tissue imaging: prospects in redox imaging and disease diagnosis.J Biomed Opt. 2005 Sep-Oct;10(5):051407. doi: 10.1117/1.2098753. J Biomed Opt. 2005. PMID: 16292944 Review.
Cited by
-
On-the-fly decoding luminescence lifetimes in the microsecond region for lanthanide-encoded suspension arrays.Nat Commun. 2014 May 6;5:3741. doi: 10.1038/ncomms4741. Nat Commun. 2014. PMID: 24796249 Free PMC article.
-
Measuring and sorting cell populations expressing isospectral fluorescent proteins with different fluorescence lifetimes.PLoS One. 2014 Oct 10;9(10):e109940. doi: 10.1371/journal.pone.0109940. eCollection 2014. PLoS One. 2014. PMID: 25302964 Free PMC article.
-
Toward the measurement of multiple fluorescence lifetimes in flow cytometry: maximizing multi-harmonic content from cells and microspheres.J Biophotonics. 2015 Nov;8(11-12):908-17. doi: 10.1002/jbio.201400115. Epub 2015 Feb 26. J Biophotonics. 2015. PMID: 25727072 Free PMC article.
-
Tempo-spectral multiplexing in flow cytometry with lifetime detection using QD-encoded polymer beads.Sci Rep. 2020 Jan 20;10(1):653. doi: 10.1038/s41598-019-56938-2. Sci Rep. 2020. PMID: 31959852 Free PMC article.
-
Toward the Development of an On-Chip Acoustic Focusing Fluorescence Lifetime Flow Cytometer.Front Phys. 2021 May;9:647985. doi: 10.3389/fphy.2021.647985. Epub 2021 May 14. Front Phys. 2021. PMID: 34386487 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

