Platelet protein disulfide isomerase is required for thrombus formation but not for hemostasis in mice
- PMID: 23788140
- PMCID: PMC3739031
- DOI: 10.1182/blood-2013-03-492504
Platelet protein disulfide isomerase is required for thrombus formation but not for hemostasis in mice
Abstract
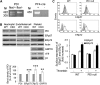
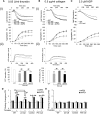
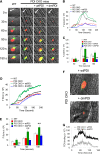
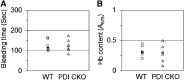
Protein disulfide isomerase (PDI) derived from intravascular cells is required for thrombus formation. However, it remains unclear whether platelet PDI contributes to the process. Using platelet-specific PDI-deficient mice, we demonstrate that PDI-null platelets have defects in aggregation and adenosine triphosphate secretion induced by thrombin, collagen, and adenosine diphosphate. Such defects were rescued by wild-type but not mutant PDI, indicating that the isomerase activity of platelet surface PDI is critical for the regulatory effect. PDI-deficient platelets expressed increased levels of intracellular ER protein 57 (ERp57) and ERp72. Platelet PDI regulated αIIbβ3 integrin activation but not P-selectin exposure, Ca(2+) mobilization, β3-talin1 interaction, or platelet spreading on immobilized fibrinogen. Inhibition of ERp57 further diminished αIIbβ3 integrin activation and aggregation of activated PDI-deficient platelets, suggesting distinct roles of PDI and ERp57 in platelet functions. We found that platelet PDI is important for thrombus formation on collagen-coated surfaces under shear. Intravital microscopy demonstrates that platelet PDI is important for platelet accumulation but not initial adhesion and fibrin generation following laser-induced arteriolar injury. Tail bleeding time in platelet-specific PDI-deficient mice were not significantly increased. Our results provide important evidence that platelet PDI is essential for thrombus formation but not for hemostasis in mice.
Figures



References
-
- Furie B, Furie BC. Mechanisms of thrombus formation. N Engl J Med. 2008;359(9):938–949. - PubMed
-
- Zucker MB, Masiello NC. Platelet aggregation caused by dithiothreitol. Thromb Haemost. 1984;51(1):119–124. - PubMed
-
- Ni H, Li A, Simonsen N, Wilkins JA. Integrin activation by dithiothreitol or Mn2+ induces a ligand-occupied conformation and exposure of a novel NH2-terminal regulatory site on the beta1 integrin chain. J Biol Chem. 1998;273(14):7981–7987. - PubMed
-
- O’Neill S, Robinson A, Deering A, Ryan M, Fitzgerald DJ, Moran N. The platelet integrin alpha IIbbeta 3 has an endogenous thiol isomerase activity. J Biol Chem. 2000;275(47):36984–36990. - PubMed
-
- Yan B, Smith JW. A redox site involved in integrin activation. J Biol Chem. 2000;275(51):39964–39972. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

