Costs of crowding for the transmission of malaria parasites
- PMID: 23789029
- PMCID: PMC3684743
- DOI: 10.1111/eva.12048
Costs of crowding for the transmission of malaria parasites
Abstract
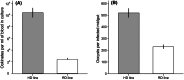
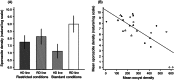
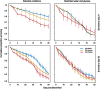
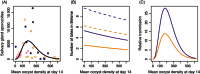
The utility of using evolutionary and ecological frameworks to understand the dynamics of infectious diseases is gaining increasing recognition. However, integrating evolutionary ecology and infectious disease epidemiology is challenging because within-host dynamics can have counterintuitive consequences for between-host transmission, especially for vector-borne parasites. A major obstacle to linking within- and between-host processes is that the drivers of the relationships between the density, virulence, and fitness of parasites are poorly understood. By experimentally manipulating the intensity of rodent malaria (Plasmodium berghei) infections in Anopheles stephensi mosquitoes under different environmental conditions, we show that parasites experience substantial density-dependent fitness costs because crowding reduces both parasite proliferation and vector survival. We then use our data to predict how interactions between parasite density and vector environmental conditions shape within-vector processes and onward disease transmission. Our model predicts that density-dependent processes can have substantial and unexpected effects on the transmission potential of vector-borne disease, which should be considered in the development and evaluation of transmission-blocking interventions.
Keywords: Anopheles stephensi; Plasmodium berghei; density dependence; disease transmission; fitness costs; life-history strategies; programmed cell death; vector-borne disease.
Figures
Similar articles
-
The dynamics of interactions between Plasmodium and the mosquito: a study of the infectivity of Plasmodium berghei and Plasmodium gallinaceum, and their transmission by Anopheles stephensi, Anopheles gambiae and Aedes aegypti.Int J Parasitol. 2003 Aug;33(9):933-43. doi: 10.1016/s0020-7519(03)00112-7. Int J Parasitol. 2003. PMID: 12906877
-
Influence of midgut microbiota in Anopheles stephensi on Plasmodium berghei infections.Malar J. 2018 Oct 25;17(1):385. doi: 10.1186/s12936-018-2535-7. Malar J. 2018. PMID: 30359252 Free PMC article.
-
Temperature impacts the environmental suitability for malaria transmission by Anopheles gambiae and Anopheles stephensi.Ecology. 2022 Aug;103(8):e3685. doi: 10.1002/ecy.3685. Epub 2022 Jun 14. Ecology. 2022. PMID: 35315521 Free PMC article.
-
Plasmodium development in Anopheles: a tale of shared resources.Trends Parasitol. 2022 Feb;38(2):124-135. doi: 10.1016/j.pt.2021.08.009. Epub 2021 Sep 18. Trends Parasitol. 2022. PMID: 34548252 Free PMC article. Review.
-
Climate, environment and transmission of malaria.Infez Med. 2016 Jun 1;24(2):93-104. Infez Med. 2016. PMID: 27367318 Review.
Cited by
-
Rethinking the extrinsic incubation period of malaria parasites.Parasit Vectors. 2018 Mar 12;11(1):178. doi: 10.1186/s13071-018-2761-4. Parasit Vectors. 2018. PMID: 29530073 Free PMC article. Review.
-
Interactions between a fungal entomopathogen and malaria parasites within a mosquito vector.Malar J. 2015 Jan 28;14:22. doi: 10.1186/s12936-014-0526-x. Malar J. 2015. PMID: 25626485 Free PMC article.
-
War and peace: social interactions in infections.Philos Trans R Soc Lond B Biol Sci. 2014 Mar 31;369(1642):20130365. doi: 10.1098/rstb.2013.0365. Print 2014 May 19. Philos Trans R Soc Lond B Biol Sci. 2014. PMID: 24686936 Free PMC article. Review.
-
Testing a non-destructive assay to track Plasmodium sporozoites in mosquitoes over time.Parasit Vectors. 2023 Nov 4;16(1):401. doi: 10.1186/s13071-023-06015-5. Parasit Vectors. 2023. PMID: 37925480 Free PMC article.
-
Streamlining sporozoite isolation from mosquitoes by leveraging the dynamics of migration to the salivary glands.Malar J. 2022 Sep 13;21(1):264. doi: 10.1186/s12936-022-04270-y. Malar J. 2022. PMID: 36100902 Free PMC article.
References
-
- Aguiler R, Dong Y, Warr E, Dimopoulos G. Anopheles infection responses; laboratory models versus field malaria transmission systems. Acta Tropica. 2005;95:285–291. - PubMed
-
- Al-Olayan EM, Williams GT, Hurd H. Apoptosis in the malaria protozoan, Plasmodium berghei: a possible mechanism for limiting intensity of infection in the mosquito. International Journal for Parasitology. 2002;32:1133–1143. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

