VEGFR1 and VEGFR2 involvement in extracellular galectin-1- and galectin-3-induced angiogenesis
- PMID: 23799140
- PMCID: PMC3684579
- DOI: 10.1371/journal.pone.0067029
VEGFR1 and VEGFR2 involvement in extracellular galectin-1- and galectin-3-induced angiogenesis
Erratum in
-
Correction: VEGFR1 and VEGFR2 Involvement in Extracellular Galectin-1- and Galectin-3-Induced Angiogenesis.PLoS One. 2023 Dec 7;18(12):e0295736. doi: 10.1371/journal.pone.0295736. eCollection 2023. PLoS One. 2023. PMID: 38060508 Free PMC article.
Abstract
Aim: Accumulating evidence suggests that extracellular galectin-1 and galectin-3 promote angiogenesis. Increased expression of galectin-1 and/or galectin-3 has been reported to be associated with tumour progression. Thus, it is critical to identify their influence on angiogenesis.
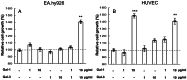
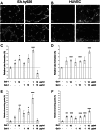
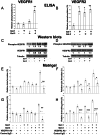
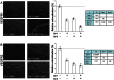
Methods: We examined the individual and combined effects of galectin-1 and galectin-3 on endothelial cell (EC) growth and tube formation using two EC lines, EA.hy926 and HUVEC. The activation of vascular endothelial growth factor receptors (VEGFR1 and VEGFR2) was determined by ELISA and Western blots. We evaluated the VEGFR1 and VEGFR2 levels in endosomes by proximity ligation assay.
Results: We observed different responses to exogenous galectins depending on the EC line. An enhanced effect on EA.hy926 cell growth and tube formation was observed when both galectins were added together. Focusing on this enhanced effect, we observed that together galectins induced the phosphorylation of both VEGFR1 and VEGFR2, whereas galectin-1 and -3 alone induced VEGFR2 phosphorylation only. In the same way, the addition of a blocking VEGFR1 antibody completely abolished the increase in tube formation induced by the combined addition of both galectins. In contrast, the addition of a blocking VEGFR2 antibody only partially inhibited this effect. Finally, the addition of both galectins induced a decrease in the VEGFR1 and VEGFR2 endocytic pools, with a significantly enhanced effect on the VEGFR1 endocytic pool. These results suggest that the combined action of galectin-1 and galectin-3 has an enhanced effect on angiogenesis via VEGFR1 activation, which could be related to a decrease in receptor endocytosis.
Conflict of interest statement
Figures

References
-
- Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144: 646–674. - PubMed
-
- Hsieh SH, Ying NW, Wu MH, Chiang WF, Hsu CL, et al. (2008) Galectin-1, a novel ligand of neuropilin-1, activates VEGFR-2 signaling and modulates the migration of vascular endothelial cells. Oncogene 27: 3746–3753. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

