Combined hepatocellular-cholangiocarcinoma in a Yellow-headed Amazon (Amazona oratrix)
- PMID: 23800973
- PMCID: PMC3942983
- DOI: 10.1292/jvms.13-0192
Combined hepatocellular-cholangiocarcinoma in a Yellow-headed Amazon (Amazona oratrix)
Abstract
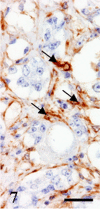
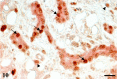
A 9-year-old male Yellow-headed Amazon (Amazona oratrix) with a history of anorexia and vomiting died of a liver tumor. The tumor consisted of neoplastic cells with hepatocellular and cholangiocellular differentiations and their intermingled areas. Neoplastic hepatocytes showed islands or trabecular growth with vacuolated eosinophilic cytoplasm. Cells showing biliary differentiation formed ducts or tubules lined by cytokeratin AE1/AE3-positive epithelia, accompanied by desmoplasia consisting of myofibroblasts reacting to α-smooth muscle actin and desmin. The tumor was diagnosed as a combined hepatocellular-cholangiocarcinoma, which is very rare in the avian.
Figures
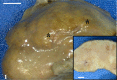

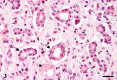

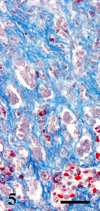
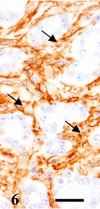


Similar articles
-
Combined hepatocellular-cholangiocarcinoma in a lesser flamingo (Phoenicopterus minor).Avian Pathol. 2010 Aug;39(4):275-8. doi: 10.1080/03079457.2010.493553. Avian Pathol. 2010. PMID: 20706883
-
Combined hepatocellular and cholangiocellular carcinoma in a dog.J Vet Med Sci. 2001 Apr;63(4):483-6. doi: 10.1292/jvms.63.483. J Vet Med Sci. 2001. PMID: 11346190
-
Combined hepatocellular carcinoma and cholangiocarcinoma in a mare.J Comp Pathol. 1997 May;116(4):409-13. doi: 10.1016/s0021-9975(97)80058-8. J Comp Pathol. 1997. PMID: 9179754
-
Spontaneously occurring hepatocellular neoplasia in adolescent cynomolgus monkeys (Macaca fascicularis).Vet Pathol. 2000 Nov;37(6):656-62. doi: 10.1354/vp.37-6-656. Vet Pathol. 2000. PMID: 11105957 Review.
-
Sarcomatoid combined hepatocellular-cholangiocarcinoma: a case report and review of literature.Int J Clin Exp Pathol. 2014 Oct 15;7(11):8290-4. eCollection 2014. Int J Clin Exp Pathol. 2014. PMID: 25550887 Free PMC article. Review.
Cited by
-
Combined hepatocellular-cholangiocarcinoma in a cow.J Vet Med Sci. 2020 Jan 17;82(1):84-88. doi: 10.1292/jvms.19-0304. Epub 2019 Dec 11. J Vet Med Sci. 2020. PMID: 31827010 Free PMC article.
References
-
- Baba A. I., Catoi C.2007. Chapter 9, Tumors of the liver and gallbladder. http://www.ncbi.nlm.nih.gov/books/NBK9548 Comparative Oncology, The Publishing House of the Romanian Academy, Bucharest. - PubMed
-
- Cullen J. M., Marison P. L., Sherman G. J., Hong X., Newbold J. E. 1990. Hepatic neoplasms in aflatoxin B1-treated, congenital duck hepatitis B virus-infected, and virus-free pekin ducks. Cancer Res. 50: 4072–4080 - PubMed
-
- Goyal D., Singh A., Sood N., Gupta K., Sood N. K. 2005. A rare case of cholangiohepatocellular carcinoma in poultry. Indian J. Vet. Pathol. 29: 52–53
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

