Control of uterine microenvironment by foxp3(+) cells facilitates embryo implantation
- PMID: 23801995
- PMCID: PMC3689029
- DOI: 10.3389/fimmu.2013.00158
Control of uterine microenvironment by foxp3(+) cells facilitates embryo implantation
Abstract
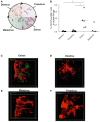
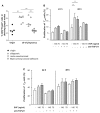
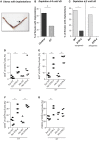
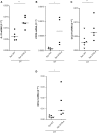
Implantation of the fertilized egg into the maternal uterus depends on the fine balance between inflammatory and anti-inflammatory processes. Whilst regulatory T cells (Tregs) are reportedly involved in protection of allogeneic fetuses against rejection by the maternal immune system, their role for pregnancy to establish, e.g., blastocyst implantation, is not clear. By using 2-photon imaging we show that Foxp3(+) cells accumulated in the mouse uterus during the receptive phase of the estrus cycle. Seminal fluid further fostered Treg expansion. Depletion of Tregs in two Foxp3.DTR-based models prior to pairing drastically impaired implantation and resulted in infiltration of activated T effector cells as well as in uterine inflammation and fibrosis in both allogeneic and syngeneic mating combinations. Genetic deletion of the homing receptor CCR7 interfered with accumulation of Tregs in the uterus and implantation indicating that homing of Tregs to the uterus was mediated by CCR7. Our results demonstrate that Tregs play a critical role in embryo implantation by preventing the development of a hostile uterine microenvironment.
Keywords: fibrosis; implantation; inflammation; pregnancy; regulatory T cells.
Figures


Similar articles
-
VIP Promotes Recruitment of Tregs to the Uterine-Placental Interface During the Peri-Implantation Period to Sustain a Tolerogenic Microenvironment.Front Immunol. 2020 Jan 8;10:2907. doi: 10.3389/fimmu.2019.02907. eCollection 2019. Front Immunol. 2020. PMID: 31969877 Free PMC article.
-
Paternal antigen-specific proliferating regulatory T cells are increased in uterine-draining lymph nodes just before implantation and in pregnant uterus just after implantation by seminal plasma-priming in allogeneic mouse pregnancy.J Reprod Immunol. 2015 Apr;108:72-82. doi: 10.1016/j.jri.2015.02.005. Epub 2015 Mar 9. J Reprod Immunol. 2015. PMID: 25817463
-
Seminal fluid regulates accumulation of FOXP3+ regulatory T cells in the preimplantation mouse uterus through expanding the FOXP3+ cell pool and CCL19-mediated recruitment.Biol Reprod. 2011 Aug;85(2):397-408. doi: 10.1095/biolreprod.110.088591. Epub 2011 Mar 9. Biol Reprod. 2011. PMID: 21389340
-
Macrophages and migratory cells in endometrium relevant to implantation.Baillieres Clin Obstet Gynaecol. 1991 Mar;5(1):25-59. doi: 10.1016/s0950-3552(05)80069-0. Baillieres Clin Obstet Gynaecol. 1991. PMID: 1855342 Review.
-
Potentiating maternal immune tolerance in pregnancy: a new challenging role for regulatory T cells.Placenta. 2014 Apr;35(4):241-8. doi: 10.1016/j.placenta.2014.02.004. Epub 2014 Feb 14. Placenta. 2014. PMID: 24581729 Review.
Cited by
-
Maternal and fetal factors that contribute to the localization of T regulatory cells during pregnancy.Am J Reprod Immunol. 2014 May;71(5):391-400. doi: 10.1111/aji.12223. Epub 2014 Mar 14. Am J Reprod Immunol. 2014. PMID: 24629013 Free PMC article.
-
The landscape of decidual immune cells at the maternal-fetal interface in parturition and preterm birth.Inflamm Res. 2025 Mar 4;74(1):44. doi: 10.1007/s00011-025-02015-6. Inflamm Res. 2025. PMID: 40038160 Free PMC article. Review.
-
Metabolic Reprogramming of Immune Cells at the Maternal-Fetal Interface and the Development of Techniques for Immunometabolism.Front Immunol. 2021 Sep 9;12:717014. doi: 10.3389/fimmu.2021.717014. eCollection 2021. Front Immunol. 2021. PMID: 34566973 Free PMC article. Review.
-
Endometrial mesenchymal stem/stromal cells: The Enigma to code messages for generation of functionally active regulatory T cells.Stem Cell Res Ther. 2021 Oct 9;12(1):536. doi: 10.1186/s13287-021-02603-3. Stem Cell Res Ther. 2021. PMID: 34627370 Free PMC article.
-
Decoding the chemokine network that links leukocytes with decidual cells and the trophoblast during early implantation.Cell Adh Migr. 2016 Mar 3;10(1-2):197-207. doi: 10.1080/19336918.2015.1135285. Epub 2016 Feb 18. Cell Adh Migr. 2016. PMID: 26891097 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

