The natural killer cell response and tumor debulking are associated with prolonged survival in recurrent glioblastoma patients receiving dendritic cells loaded with autologous tumor lysates
- PMID: 23802079
- PMCID: PMC3661164
- DOI: 10.4161/onci.23401
The natural killer cell response and tumor debulking are associated with prolonged survival in recurrent glioblastoma patients receiving dendritic cells loaded with autologous tumor lysates
Abstract
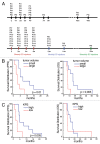
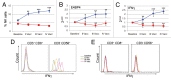
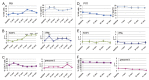
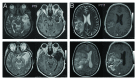
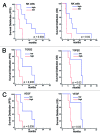
Recurrent glioblastomas (GBs) are highly aggressive tumors associated with a 6-8 mo survival rate. In this study, we evaluated the possible benefits of an immunotherapeutic strategy based on mature dendritic cells (DCs) loaded with autologous tumor-cell lysates in 15 patients affected by recurrent GB. The median progression-free survival (PFS) of this patient cohort was 4.4 mo, and the median overall survival (OS) was 8.0 mo. Patients with small tumors at the time of the first vaccination (< 20 cm3; n = 8) had significantly longer PFS and OS than the other patients (6.0 vs. 3.0 mo, p = 0.01; and 16.5 vs. 7.0 mo, p = 0.003, respectively). CD8+ T cells, CD56+ natural killer (NK) cells and other immune parameters, such as the levels of transforming growth factor β, vascular endothelial growth factor, interleukin-12 and interferon γ (IFNγ), were measured in the peripheral blood and serum of patients before and after immunization, which enabled us to obtain a vaccination/baseline ratio (V/B ratio). An increased V/B ratio for NK cells, but not CD8+ T cells, was significantly associated with prolonged PFS and OS. Patients exhibiting NK-cell responses were characterized by high levels of circulating IFNγ and E4BP4, an NK-cell transcription factor. Furthermore, the NK cell V/B ratio was inversely correlated with the TGFβ2 and VEGF V/B ratios. These results suggest that tumor-loaded DCs may increase the survival rate of patients with recurrent GB after effective tumor debulking, and emphasize the role of the NK-cell response in this therapeutic setting.
Keywords: IFNγ; NK cells; dendritic cells; glioblastoma; immunotherapy.
Figures
Similar articles
-
Predictors of Response to Autologous Dendritic Cell Therapy in Glioblastoma Multiforme.Front Immunol. 2018 May 29;9:727. doi: 10.3389/fimmu.2018.00727. eCollection 2018. Front Immunol. 2018. PMID: 29910795 Free PMC article. Clinical Trial.
-
Cytokine-induced killer cell therapy for modulating regulatory T cells in patients with non-small cell lung cancer.Exp Ther Med. 2017 Jul;14(1):831-840. doi: 10.3892/etm.2017.4562. Epub 2017 Jun 8. Exp Ther Med. 2017. PMID: 28673007 Free PMC article.
-
Clinically feasible approaches to potentiating cancer cell-based immunotherapies.Hum Vaccin Immunother. 2015;11(4):851-69. doi: 10.1080/21645515.2015.1009814. Hum Vaccin Immunother. 2015. PMID: 25933181 Free PMC article. Review.
-
Natural killer T cell activation overcomes immunosuppression to enhance clearance of postsurgical breast cancer metastasis in mice.Oncoimmunology. 2015 Jan 22;4(3):e995562. doi: 10.1080/2162402X.2014.995562. eCollection 2015 Mar. Oncoimmunology. 2015. PMID: 25949924 Free PMC article.
-
CAR-Engineered NK Cells for the Treatment of Glioblastoma: Turning Innate Effectors Into Precision Tools for Cancer Immunotherapy.Front Immunol. 2019 Nov 14;10:2683. doi: 10.3389/fimmu.2019.02683. eCollection 2019. Front Immunol. 2019. PMID: 31798595 Free PMC article. Review.
Cited by
-
Imaging-based Biomarkers for Predicting and Evaluating Cancer Immunotherapy Response.Radiol Imaging Cancer. 2019 Nov 29;1(2):e190031. doi: 10.1148/rycan.2019190031. eCollection 2019 Nov. Radiol Imaging Cancer. 2019. PMID: 33778682 Free PMC article. Review.
-
Opportunities in immunotherapy of ovarian cancer.Ann Oncol. 2016 Apr;27 Suppl 1(Suppl 1):i11-i15. doi: 10.1093/annonc/mdw084. Ann Oncol. 2016. PMID: 27141063 Free PMC article. Review.
-
Regulation of MHC I Molecules in Glioblastoma Cells and the Sensitizing of NK Cells.Pharmaceuticals (Basel). 2021 Mar 8;14(3):236. doi: 10.3390/ph14030236. Pharmaceuticals (Basel). 2021. PMID: 33800301 Free PMC article. Review.
-
The multidrug-resistance transporter Abcc3 protects NK cells from chemotherapy in a murine model of malignant glioma.Oncoimmunology. 2015 Oct 29;5(5):e1108513. doi: 10.1080/2162402X.2015.1108513. eCollection 2016 May. Oncoimmunology. 2015. PMID: 27467914 Free PMC article.
-
Interleukin-15 Dendritic Cells Harness NK Cell Cytotoxic Effector Function in a Contact- and IL-15-Dependent Manner.PLoS One. 2015 May 7;10(5):e0123340. doi: 10.1371/journal.pone.0123340. eCollection 2015. PLoS One. 2015. PMID: 25951230 Free PMC article.
References
-
- Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, et al. European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups. National Cancer Institute of Canada Clinical Trials Group Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352:987–96. doi: 10.1056/NEJMoa043330. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
