Immunological and virological analyses of rhesus macaques immunized with chimpanzee adenoviruses expressing the simian immunodeficiency virus Gag/Tat fusion protein and challenged intrarectally with repeated low doses of SIVmac
- PMID: 23804645
- PMCID: PMC3754116
- DOI: 10.1128/JVI.01456-13
Immunological and virological analyses of rhesus macaques immunized with chimpanzee adenoviruses expressing the simian immunodeficiency virus Gag/Tat fusion protein and challenged intrarectally with repeated low doses of SIVmac
Abstract
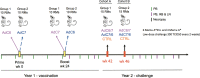
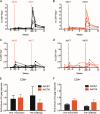
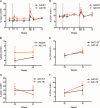
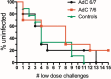
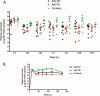
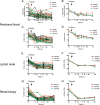
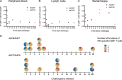
Human adenovirus (AdHu)-based candidate AIDS vaccine can provide protection from simian immunodeficiency virus (SIV) transmission and disease progression. However, their potential use may be limited by widespread preexisting immunity to the vector. In contrast, preexisting immunity to chimpanzee adenoviruses (AdC) is relatively rare. In this study, we utilized two regimens of prime-boost immunizations with AdC serotype SAd-V23 (also called AdC6) and SAd-V24 (also called AdC7) expressing SIV Gag/Tat to test their immunogenicity and ability to protect rhesus macaques (RMs) from a repeated low-dose SIVmac239 challenge. Both AdC6 followed by AdC7 (AdC6/7) and AdC7 followed by AdC6 (AdC7/6) induced robust SIV Gag/Tat-specific T cell responses as measured by tetramer staining and functional assays. However, no significant protection from SIV transmission was observed in either AdC7/6- or AdC7/6-vaccinated RMs. Interestingly, in the RMs showing breakthrough infections, AdC7/6-SIV immunization was associated with a transient but significant (P = 0.035 at day 90 and P = 0.033 at day 120 postinfection) reduction in the setpoint viral load compared to unvaccinated controls. None of the measured immunological markers (i.e., number or functionality of SIV-specific CD8(+) and CD4(+) T cell responses and level of activated and/or CCR5(+) CD4(+) target cells) at the time of challenge correlated with protection from SIV transmission in the AdC-SIV-vaccinated RMs. The robust immunogenicity observed in all AdC-immunized RMs and the transient signal of protection from SIV replication exhibited by AdC7/6-vaccinated RMs even in the absence of any envelope immunogen suggest that AdC-based vectors may represent a promising platform for candidate AIDS vaccines.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

