Alzheimer's amyloid-β oligomers rescue cellular prion protein induced tau reduction via the Fyn pathway
- PMID: 23805846
- PMCID: PMC3778425
- DOI: 10.1021/cn400085q
Alzheimer's amyloid-β oligomers rescue cellular prion protein induced tau reduction via the Fyn pathway
Abstract
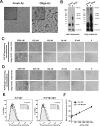
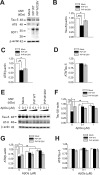
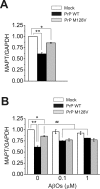
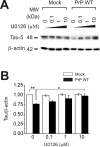
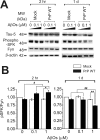
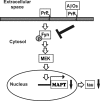
Amyloid-β (Aβ) and tau are the pathogenic hallmarks in Alzheimer's disease (AD). Aβ oligomers are considered the actual toxic entities, and the toxicity relies on the presence of tau. Recently, Aβ oligomers have been shown to specifically interact with cellular prion protein (PrP(C)) where the role of PrP(C) in AD is still not fully understood. To investigate the downstream mechanism of PrP(C) and Aβ oligomer interaction and their possible relationships to tau, we examined tau expression in human neuroblastoma BE(2)-C cells transfected with murine PrP(C) and studied the effect under Aβ oligomer treatment. By Western blotting, we found that PrP(C) overexpression down-regulated tau protein and Aβ oligomer binding alleviated the tau reduction induced by wild type but not M128V PrP(C), the high AD risk polymorphic allele in human prion gene. PrP(C) lacking the Aβ oligomer binding site was incapable of rescuing the level of tau reduction. Quantitative RT-PCR showed the PrP(C) effect was attributed to tau reduction at the transcription level. Treatment with Fyn pathway inhibitors, Fyn kinase inhibitor PP2 and MEK inhibitor U0126, reversed the PrP(C)-induced tau reduction and Aβ oligomer treatment modulated Fyn kinase activity. The results suggested Fyn pathway regulated Aβ-PrP(C)-tau signaling. Overall, our results demonstrated that PrP(C) down-regulated tau via the Fyn pathway and the effect can be regulated by Aβ oligomers. Our study facilitated the understanding of molecular mechanisms among PrP(C), tau, and Aβ oligomers.
Figures


References
-
- Lesne S.; Koh M. T.; Kotilinek L.; Kayed R.; Glabe C. G.; Yang A.; Gallagher M.; Ashe K. H. (2006) A specific amyloid-beta protein assembly in the brain impairs memory. Nature 440(7082), 352–35. - PubMed
- Shankar G. M.; Li S.; Mehta T. H.; Garcia-Munoz A.; Shepardson N. E.; Smith I.; Brett F. M.; Farrell M. A.; Rowan M. J.; Lemere C. A.; Regan C. M.; Walsh D. M.; Sabatini B. L.; Selkoe D. J. (2008) Amyloid-beta protein dimers isolated directly from Alzheimer’s brains impair synaptic plasticity and memory. Nat. Med. 14(8), 837–842. - PMC - PubMed
-
- Haass C.; Selkoe D. J. (2007) Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer’s amyloid beta-peptide. Nat. Rev. Mol. Cell Biol. 8(2), 101–112. - PubMed
-
- Avila J.; Lucas J. J.; Perez M.; Hernandez F. (2004) Role of tau protein in both physiological and pathological conditions. Physiol. Rev. 84(2), 361–384. - PubMed
-
- Roberson E. D.; Scearce-Levie K.; Palop J. J.; Yan F.; Cheng I. H.; Wu T.; Gerstein H.; Yu G. Q.; Mucke L. (2007) Reducing endogenous tau ameliorates amyloid beta-induced deficits in an Alzheimer’s disease mouse model. Science 316(5825), 750–754. - PubMed
- Vossel K. A.; Zhang K.; Brodbeck J.; Daub A. C.; Sharma P.; Finkbeiner S.; Cui B.; Mucke L. (2010) Tau reduction prevents Abeta-induced defects in axonal transport. Science 330(6001), 198. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

