A pilot study on quality of artesunate and amodiaquine tablets used in the fishing community of Tema, Ghana
- PMID: 23809666
- PMCID: PMC3722045
- DOI: 10.1186/1475-2875-12-220
A pilot study on quality of artesunate and amodiaquine tablets used in the fishing community of Tema, Ghana
Abstract
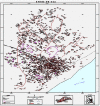
Background: The ineffectiveness of artesunate and amodiaquine tablets in malaria treatment remains a health burden to WHO and governments of malaria-endemic countries, including Ghana. The proliferation of illegitimate anti-malarial drugs and its use by patients is of primary concern to international and local drug regulatory agencies because such drugs are known to contribute to the development of the malaria-resistant parasites in humans. No data exist on quality of these drugs in the fishing village communities in Ghana although the villagers are likely users of such drugs. A pilot study on the quality of anti-malarial tablets in circulation during the major fishing season at a malarious fishing village located along the coast of Tema in southern Ghana was determined.
Methods: Blisterpacks of anti-malarial tablets were randomly sampled. The International Pharmacopoeia and Global Pharma Health Fund Minilab protocols were used to assess the quality of anti-malarial tablets per blisterpacks allegedly manufactured by Guilin Pharmaceutical Co Ltd, China (GPCL) and Letap Pharmaceuticals Ltd, Ghana (LPL) and sold in chemical sales outlets at Kpone-on-Sea. Ferric chloride and cobaltous thiocyanate tests confirmed the presence of active ingredients in the tablets. A confirmatory test for the active ingredient was achieved with artesunate (ICRS1409) and amodiaquine (ICRS0209) reference standards. A high performance liquid chromatography analysis confirmed the amount of artesunate found in tablets.
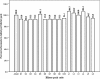
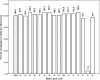
Results: Based on the International Pharmacopoeia acceptable range of 96/98 to 102% for genuine artesunate per tablet, 10% [relative standard deviation (RSD): 3.2%] of field-selected artesunate blisterpack per tablets manufactured by GPCL, and 50% (RSD: 5.1%) of a similar package per tablet by LPL, passed the titrimetric test. However, 100% (RSD: 2.2%) of amodiaquine blisterpack per tablet by GPCL were found to be within the International Pharmacopeia acceptable range of 90 to 110% for genuine amodiaquine in tablet, whilst 17% of a similar package per tablet by LPL failed spectrophotometric testing.
Conclusion: Inadequate amounts of artesunate and amodiaquine detected in the tablets suggest that both pharmaceutical companies may not be following recommended drug formulation procedures, or the active pharmaceutical ingredients might have been degraded by improper storage conditions. Thus, drugs being sold at Kpone-on-Sea, Ghana may likely be classified as substandard drugs and not suitable for malaria treatment.
Figures
Similar articles
-
Substandard artemisinin-based antimalarial medicines in licensed retail pharmaceutical outlets in Ghana.J Vector Borne Dis. 2012 Sep;49(3):131-9. J Vector Borne Dis. 2012. PMID: 23135006
-
Capillary electrophoresis for the assay of fixed-dose combination tablets of artesunate and amodiaquine.Malar J. 2012 May 3;11:149. doi: 10.1186/1475-2875-11-149. Malar J. 2012. PMID: 22554086 Free PMC article.
-
A New Handheld Device for the Detection of Falsified Medicines: Demonstration on Falsified Artemisinin-Based Therapies from the Field.Am J Trop Med Hyg. 2017 May;96(5):1117-1123. doi: 10.4269/ajtmh.16-0904. Epub 2017 May 13. Am J Trop Med Hyg. 2017. PMID: 28219992 Free PMC article.
-
[Combined antimalarial therapy using artemisinin].Parassitologia. 2004 Jun;46(1-2):85-7. Parassitologia. 2004. PMID: 15305693 Review. Italian.
-
Pyronaridine-artesunate for treating uncomplicated Plasmodium falciparum malaria.Cochrane Database Syst Rev. 2019 Jan 8;1(1):CD006404. doi: 10.1002/14651858.CD006404.pub3. Cochrane Database Syst Rev. 2019. Update in: Cochrane Database Syst Rev. 2022 Jun 21;6:CD006404. doi: 10.1002/14651858.CD006404.pub4. PMID: 30620055 Free PMC article. Updated.
Cited by
-
Are we doing enough to prevent poor-quality antimalarial medicines in the developing world?BMC Public Health. 2018 May 15;18(1):630. doi: 10.1186/s12889-018-5521-7. BMC Public Health. 2018. PMID: 29764407 Free PMC article.
-
Anti-malarial medicine quality field studies and surveys: a systematic review of screening technologies used and reporting of findings.Malar J. 2017 May 15;16(1):197. doi: 10.1186/s12936-017-1852-6. Malar J. 2017. PMID: 28506234 Free PMC article.
-
Assessing the Impact of Substandard and Falsified Antimalarials in Benin.Am J Trop Med Hyg. 2021 Nov 8;106(6):1770-7. doi: 10.4269/ajtmh.21-0450. Online ahead of print. Am J Trop Med Hyg. 2021. PMID: 34749311 Free PMC article.
-
Quality of antimalarials at the epicenter of antimalarial drug resistance: results from an overt and mystery client survey in Cambodia.Am J Trop Med Hyg. 2015 Jun;92(6 Suppl):39-50. doi: 10.4269/ajtmh.14-0391. Epub 2015 Apr 20. Am J Trop Med Hyg. 2015. PMID: 25897063 Free PMC article.
-
The economic impact of substandard and falsified antimalarial medications in Nigeria.PLoS One. 2019 Aug 15;14(8):e0217910. doi: 10.1371/journal.pone.0217910. eCollection 2019. PLoS One. 2019. PMID: 31415560 Free PMC article.
References
-
- Goodman LS, Gilman AG. The pharmacological basis of therapeutics. 10. New York: McGraw-Hill; 2001. p. 1069.
-
- WHO. Malaria. [ http://www.who.int/tdr/diseases-topics/malaria/en/]. Accessed: 02.06.2012.
-
- WHO. Counterfeit drugs. Guidelines for the development of measures to combat counterfeit drugs. WHO/EDM/GSM/99.1. Geneva: World Health Organization; 1999. [ http://apps.who.int/medicinedocs/pdf/h1456e/h1456e.pdf]. Accessed: 03.24.2012.
-
- WHO. World malaria report 2008. Geneva: World Health Organization; 2008. [ http://www.who.int/malaria/wmr2008]. Accessed: 06.02.2010.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

