Molecular classification of prostate cancer progression: foundation for marker-driven treatment of prostate cancer
- PMID: 23811619
- PMCID: PMC3926428
- DOI: 10.1158/2159-8290.CD-12-0460
Molecular classification of prostate cancer progression: foundation for marker-driven treatment of prostate cancer
Abstract
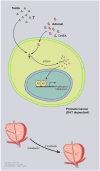
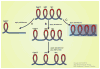
Recently, many therapeutic agents for prostate cancer have been approved that target the androgen receptor and/or the prostate tumor microenvironment. Each of these therapies has modestly increased patient survival. A better understanding of when in the course of prostate cancer progression specific therapies should be applied, and of what biomarkers would indicate when resistance arises, would almost certainly improve survival due to these therapies. Thus, applying the armamentarium of therapeutic agents in the right sequences in the right combination at the right time is a major goal in prostate cancer treatment. For this to occur, an understanding of prostate cancer evolution during progression is required. In this review, we discuss the current understanding of prostate cancer progression, but challenge the prevailing view by proposing a new model of prostate cancer progression, with the goal of improving biologic classification and treatment strategies. We use this model to discuss how integrating clinical and basic understanding of prostate cancer will lead to better implementation of molecularly targeted therapeutics and improve patient survival.
Figures


References
-
- Gleason DF. Classification of prostatic carcinomas. Cancer Chemother Rep. 1966;50:125–8. - PubMed
-
- Gleason DF, Mellinger GT. Prediction of prognosis for prostatic adenocarcinoma by combined histological grading and clinical staging. The Journal of urology. 1974;111:58–64. - PubMed
-
- Ryan CJ, Smith A, Lal P, Satagopan J, Reuter V, Scardino P, et al. Persistent prostate-specific antigen expression after neoadjuvant androgen depletion: an early predictor of relapse or incomplete androgen suppression. Urology. 2006;68:834–9. - PubMed
-
- McKenney JK, Simko J, Bonham M, True LD, Troyer D, Hawley S, et al. The potential impact of reproducibility of Gleason grading in men with early stage prostate cancer managed by active surveillance: a multi-institutional study. The Journal of urology. 2011;186:465–9. - PubMed
-
- Gravis G, Fizazi K, Joly F, Oudard S, Priou F, Esterni B, et al. Androgen-deprivation therapy alone or with docetaxel in non-castrate metastatic prostate cancer (GETUG-AFU 15): a randomised, open-label, phase 3 trial. The lancet oncology. 2013;14:149–58. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

