The mitochondrial-targeted compound SS-31 re-energizes ischemic mitochondria by interacting with cardiolipin
- PMID: 23813215
- PMCID: PMC3736700
- DOI: 10.1681/ASN.2012121216
The mitochondrial-targeted compound SS-31 re-energizes ischemic mitochondria by interacting with cardiolipin
Abstract
Ischemia causes AKI as a result of ATP depletion, and rapid recovery of ATP on reperfusion is important to minimize tissue damage. ATP recovery is often delayed, however, because ischemia destroys the mitochondrial cristae membranes required for mitochondrial ATP synthesis. The mitochondria-targeted compound SS-31 accelerates ATP recovery after ischemia and reduces AKI, but its mechanism of action remains unclear. Here, we used a polarity-sensitive fluorescent analog of SS-31 to demonstrate that SS-31 binds with high affinity to cardiolipin, an anionic phospholipid expressed on the inner mitochondrial membrane that is required for cristae formation. In addition, the SS-31/cardiolipin complex inhibited cytochrome c peroxidase activity, which catalyzes cardiolipin peroxidation and results in mitochondrial damage during ischemia, by protecting its heme iron. Pretreatment of rats with SS-31 protected cristae membranes during renal ischemia and prevented mitochondrial swelling. Prompt recovery of ATP on reperfusion led to rapid repair of ATP-dependent processes, such as restoration of the actin cytoskeleton and cell polarity. Rapid recovery of ATP also inhibited apoptosis, protected tubular barrier function, and mitigated renal dysfunction. In conclusion, SS-31, which is currently in clinical trials for ischemia-reperfusion injury, protects mitochondrial cristae by interacting with cardiolipin on the inner mitochondrial membrane.
Figures

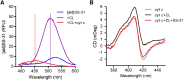


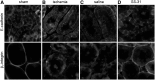
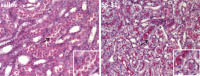


Comment in
-
Maintaining mitochondrial morphology in AKI: looks matter.J Am Soc Nephrol. 2013 Jul;24(8):1185-7. doi: 10.1681/ASN.2013050519. Epub 2013 Jul 11. J Am Soc Nephrol. 2013. PMID: 23813213 Free PMC article. No abstract available.
References
-
- Bon D, Chatauret N, Giraud S, Thuillier R, Favreau F, Hauet T: New strategies to optimize kidney recovery and preservation in transplantation. Nat Rev Nephrol 8: 339–347, 2012 - PubMed
-
- Chapman JR, O’Connell PJ, Nankivell BJ: Chronic renal allograft dysfunction. J Am Soc Nephrol 16: 3015–3026, 2005 - PubMed
-
- Atkinson SJ, Hosford MA, Molitoris BA: Mechanism of actin polymerization in cellular ATP depletion. J Biol Chem 279: 5194–5199, 2004 - PubMed
-
- Sharfuddin AA, Molitoris BA: Pathophysiology of ischemic acute kidney injury. Nat Rev Nephrol 7: 189–200, 2011 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

