Alpha-tocopherol succinate- and AMD3100-mobilized progenitors mitigate radiation combined injury in mice
- PMID: 23814114
- PMCID: PMC3885121
- DOI: 10.1093/jrr/rrt088
Alpha-tocopherol succinate- and AMD3100-mobilized progenitors mitigate radiation combined injury in mice
Abstract
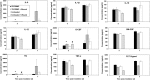
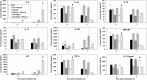
The purpose of this study was to elucidate the role of alpha-tocopherol succinate (TS)- and AMD3100-mobilized progenitors in mitigating combined injury associated with acute radiation exposure in combination with secondary physical wounding. CD2F1 mice were exposed to high doses of cobalt-60 gamma-radiation and then transfused intravenously with 5 million peripheral blood mononuclear cells (PBMCs) from TS- and AMD3100-injected mice after irradiation. Within 1 h after irradiation, mice were exposed to secondary wounding. Mice were observed for 30 d after irradiation and cytokine analysis was conducted by multiplex Luminex assay at various time-points after irradiation and wounding. Our results initially demonstrated that transfusion of TS-mobilized progenitors from normal mice enhanced survival of acutely irradiated mice exposed 24 h prior to transfusion to supralethal doses (11.5-12.5 Gy) of (60)Co gamma-radiation. Subsequently, comparable transfusions of TS-mobilized progenitors were shown to significantly mitigate severe combined injuries in acutely irradiated mice. TS administered 24 h before irradiation was able to protect mice against combined injury as well. Cytokine results demonstrated that wounding modulates irradiation-induced cytokines. This study further supports the conclusion that the infusion of TS-mobilized progenitor-containing PBMCs acts as a bridging therapy in radiation-combined-injury mice. We suggest that this novel bridging therapeutic approach involving the infusion of TS-mobilized hematopoietic progenitors following acute radiation exposure or combined injury might be applicable to humans.
Keywords: cytokines; gamma-radiation; hematopoietic progenitor cells; mice; transfusion.
Figures
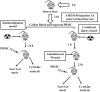
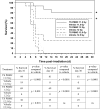

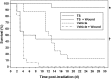
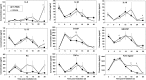
Similar articles
-
α-Tocopherol succinate- and AMD3100-mobilized progenitors mitigate radiation-induced gastrointestinal injury in mice.Exp Hematol. 2012 May;40(5):407-17. doi: 10.1016/j.exphem.2012.01.005. Epub 2012 Jan 10. Exp Hematol. 2012. PMID: 22240608
-
Alpha-tocopherol succinate-mobilized progenitors improve intestinal integrity after whole body irradiation.Int J Radiat Biol. 2013 May;89(5):334-45. doi: 10.3109/09553002.2013.762137. Epub 2013 Feb 1. Int J Radiat Biol. 2013. PMID: 23270472
-
Preclinical development of a bridging therapy for radiation casualties: appropriate for high risk personnel.Health Phys. 2014 Jun;106(6):689-98. doi: 10.1097/HP.0000000000000089. Health Phys. 2014. PMID: 24776901
-
Mobilized progenitor cells as a bridging therapy for radiation casualties: a brief review of tocopherol succinate-based approaches.Int Immunopharmacol. 2011 Jul;11(7):842-47. doi: 10.1016/j.intimp.2011.01.017. Epub 2011 Feb 3. Int Immunopharmacol. 2011. PMID: 21296698 Review.
-
Ionizing radiation-induced altered microRNA expression as biomarkers for assessing acute radiation injury.Expert Rev Mol Diagn. 2017 Oct;17(10):871-874. doi: 10.1080/14737159.2017.1366316. Epub 2017 Aug 14. Expert Rev Mol Diagn. 2017. PMID: 28792262 Free PMC article. Review. No abstract available.
Cited by
-
New approaches to radiation protection.Front Oncol. 2015 Jan 20;4:381. doi: 10.3389/fonc.2014.00381. eCollection 2014. Front Oncol. 2015. PMID: 25653923 Free PMC article. Review.
-
Protective effects of seabuckthorn pulp and seed oils against radiation-induced acute intestinal injury.J Radiat Res. 2017 Jan;58(1):24-32. doi: 10.1093/jrr/rrw069. Epub 2016 Jul 15. J Radiat Res. 2017. PMID: 27422938 Free PMC article.
-
Emerging targets for radioprotection and radiosensitization in radiotherapy.Tumour Biol. 2016 Sep;37(9):11589-11609. doi: 10.1007/s13277-016-5117-8. Epub 2016 Jun 19. Tumour Biol. 2016. PMID: 27318945 Review.
-
Metabolomic Analysis Reveals Unique Biochemical Signatures Associated with Protection from Radiation Induced Lung Injury by Lack of cd47 Receptor Gene Expression.Metabolites. 2019 Oct 8;9(10):218. doi: 10.3390/metabo9100218. Metabolites. 2019. PMID: 31597291 Free PMC article.
-
Personalized Radioproteomics: Identification of a Protein Biomarker Signature for Preemptive Rescue by Tocopherol Succinate in CD34+ Irradiated Progenitor Cells Isolated from a Healthy Control Donor.J Proteomics Bioinform. 2015 Feb;8(2):23-30. doi: 10.4172/jpb.1000349. Epub 2015 Jan 28. J Proteomics Bioinform. 2015. PMID: 27087761 Free PMC article.
References
-
- Carter AB, May MM, Perry WJ. The day after: action following a nuclear blast in a U.S. city. Washington Quarterly. 2007;30:19–32.
-
- Pellmar TC, Rockwell S. Priority list of research areas for radiological nuclear threat countermeasures. Radiat Res. 2005;163:115–23. - PubMed
-
- Benjamin GC, McGeary M, McCutchen SR. Workshop Report. Washington, DC: The National Academies Press; 2009. Assessing medical preparedness to respond to a terrorist nuclear event. - PubMed
-
- Kishi HS. Effects of the “special bomb”: recollections of a neurosurgeon in Hiroshima, August 8–15, 1945. Neurosurgery. 2000;47:441–5. discussion 45–6. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

